Reactions of N, O- and N, S-Azoles and -Azolines with Ozone: Kinetics and Mechanisms
- PMID: 40577279
- PMCID: PMC12269093
- DOI: 10.1021/acs.est.5c01323
Reactions of N, O- and N, S-Azoles and -Azolines with Ozone: Kinetics and Mechanisms
Abstract
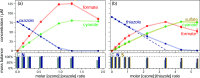
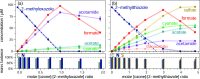
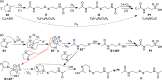
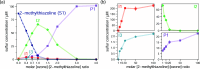
N,O- and N,S-azoles and -azolines are common functional groups in pharmaceuticals, agrochemicals and natural products. Their fate during ozone-based water treatment processes is unknown due to a lack of kinetic and mechanistic information on their reactions with ozone. Apparent second-order rate constants kO3 of 12 model compounds were determined at pH 7: oxazoles react 2 orders of magnitude faster (kO3 = 9 × 102-5 × 104 M-1s-1, depending on their substituents) than thiazoles (kO3 = 1 × 101-2 × 103 M-1s-1). The low kO3 of thiazoles limits their degradability during ozonation. Only small yields of reactive oxygen species (•OH, H2O2, and 1O2) were observed during ozonation of oxazoles, suggesting that all oxygen atoms from ozone are incorporated into the products. Oxazoles and thiazoles react initially by a Criegee-type reaction at the C=C double bond, followed by two reaction branches, leading to two observed product groups: (1) carboxylates and cyanate; (2) formate, amide and CO2. For thiazoles, thiocarboxylic acids were identified as intermediates, reacting further to sulfate and carboxylic acids, forming 1O2. The nonaromatic 2-methyloxazoline is unreactive toward ozone. 2-Methylthiazoline reacts fast (kO3 = 2 × 104 M-1s-1), forming 1O2, leading to ring-opening and formation of dimerization products which react further to N-acetyltaurine. These results enhance the understanding of the ozone reactivity of heterocycles and help predict transformation product formation.
Keywords: aromatic and nonaromatic N-heterocycles; oxazole; oxazoline; ozonation; thiazole; thiazoline.
Figures

Similar articles
-
Sexual Harassment and Prevention Training.2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 36508513 Free Books & Documents.
-
Comparison of Two Modern Survival Prediction Tools, SORG-MLA and METSSS, in Patients With Symptomatic Long-bone Metastases Who Underwent Local Treatment With Surgery Followed by Radiotherapy and With Radiotherapy Alone.Clin Orthop Relat Res. 2024 Dec 1;482(12):2193-2208. doi: 10.1097/CORR.0000000000003185. Epub 2024 Jul 23. Clin Orthop Relat Res. 2024. PMID: 39051924
-
Oral antifungal medication for toenail onychomycosis.Cochrane Database Syst Rev. 2017 Jul 14;7(7):CD010031. doi: 10.1002/14651858.CD010031.pub2. Cochrane Database Syst Rev. 2017. PMID: 28707751 Free PMC article.
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of paclitaxel, docetaxel, gemcitabine and vinorelbine in non-small-cell lung cancer.Health Technol Assess. 2001;5(32):1-195. doi: 10.3310/hta5320. Health Technol Assess. 2001. PMID: 12065068
-
Comparing the abatement of pharmaceuticals and their human metabolites in wastewater treatment plants - Insights from biological and advanced treatment stages.Water Res. 2025 Jun 24;285:123983. doi: 10.1016/j.watres.2025.123983. Online ahead of print. Water Res. 2025. PMID: 40602065
References
-
- von Sonntag, C. ; von Gunten, U. . Chemistry of Ozone in Water and Wastewater Treatment: From Basic Principles to Applications; IWA Publishing: London, 2012. 10.2166/9781780400839. - DOI
-
- Gerrity D., Pecson B., Trussell R. S., Trussell R. R.. Potable Reuse Treatment Trains throughout the World. J. Water Supply: Res. Technol.-Aqua. 2013;62(6):321–338. doi: 10.2166/AQUA.2013.041. - DOI
-
- Kienle C., Werner I., Fischer S., Lüthi C., Schifferli A., Besselink H., Langer M., McArdell C. S., Vermeirssen E. L. M.. Evaluation of a Full-Scale Wastewater Treatment Plant with Ozonation and Different Post-Treatments Using a Broad Range of in Vitro and in Vivo Bioassays. Water Res. 2022;212:118084. doi: 10.1016/j.watres.2022.118084. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

