Deep Learning Model for Natural Language to Assess Effectiveness of Patients With Non-Muscle Invasive Bladder Cancer Receiving Intravesical Bacillus Calmette-Guérin Therapy
- PMID: 40577661
- PMCID: PMC12233173
- DOI: 10.1200/CCI-24-00249
Deep Learning Model for Natural Language to Assess Effectiveness of Patients With Non-Muscle Invasive Bladder Cancer Receiving Intravesical Bacillus Calmette-Guérin Therapy
Abstract
Purpose: Collecting information on clinical outcomes (recurrence/progression) from complex treatment courses in non-muscle invasive bladder cancer (NMIBC) is challenging and time-consuming. We developed a deep learning natural language processing model to assess outcomes in patients with NMIBC using vast data from electronic health records (EHRs).
Methods: This retrospective study analyzed data from Japanese adults with NMIBC who started Bacillus Calmette-Guérin (BCG) induction therapy between April 2016 and June 2022. A Bidirectional Encoder Representations from Transformers (BERT) model was trained to classify outcomes, supported by human review for past history records. The model's performance was assessed by precision, recall, and F1 scores. We compared the effectiveness of BCG therapy between completion (patients who completed therapy) and non-completion groups.
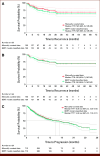
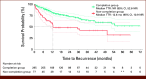
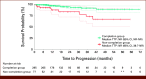
Results: Of 372 patients studied, 79.3% and 20.7% were in the completion group and the non-completion group, respectively. The final BERT model achieved average F1 scores of 0.91 and 0.98 for time to recurrence (TTR), and 0.74 and 0.94 for time to progression (TTP) before and after human support, respectively. The hazard ratio for TTR in BCG completion versus non-completion groups was 0.40 (95% CI, 0.26 to 0.62) by a multivariate Cox proportional hazard model and 0.41 (95% CI, 0.26 to 0.63) by inverse probability of treatment weighting.
Conclusion: The developed model could compare the clinical outcomes between treatments in patients with NMIBC using EHRs. Human support, although required, was needed in only 10% documents and was deemed feasible. The model was able to demonstrate the difference in TTR and TTP between BCG completion and non-completion groups.
Conflict of interest statement
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated unless otherwise noted. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to
Open Payments is a public database containing information reported by companies about payments made to US-licensed physicians (
Figures
References
-
- Gontero P, Birtle A, Compérat E, et al. EAU Guidelines on Non-Muscle-Invasive Bladder Cancer. 2024. https://d56bochluxqnz.cloudfront.net/documents/full-guideline/EAU-Guidel...
-
- Matsumoto H, Shiraishi K, Azuma H, et al. Clinical practice guidelines for bladder cancer 2019 update by the Japanese Urological Association: Summary of the revision. Int J Urol. 2020;27:702–709. - PubMed
-
- Holzbeierlein JM, Bixler BR, Buckley DI, et al. Diagnosis and treatment of non-muscle invasive bladder cancer: AUA/SUO guideline: 2024 amendment. J Urol. 2024;211:533–538. - PubMed
-
- Abou Chakra M, Luo Y, Duquesne I, et al. Update on the mechanism of action of intravesical BCG therapy to treat non-muscle-invasive bladder cancer. Front Biosci (Landmark Ed) 2024;29:295. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

