Protocol for patterning competency assay in regenerative axolotl limb cells using the competency accessory limb model
- PMID: 40580475
- PMCID: PMC12266493
- DOI: 10.1016/j.xpro.2025.103920
Protocol for patterning competency assay in regenerative axolotl limb cells using the competency accessory limb model
Abstract
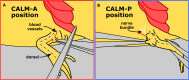
A key question in limb regeneration is how axolotl limb cells become competent to specific types of regeneration signals. Here, we present the competency accessory limb model (CALM), a regenerative assay that assesses whether limb cells have achieved the ability to respond to patterning signals. We describe the steps for positioning a wound, deviating the nerve into a limb wound site, and injecting retinoic acid. We also detail the harvest and analyses in both anterior and posterior CALMs. For complete details on the use and execution of this protocol, please refer to Raymond et al.1.
Keywords: Cell Biology; Developmental biology; Model Organisms; Molecular Biology.
Copyright © 2025 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures





References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

