Low-dose radiotherapy enhances the efficacy of PD-L1 blockade and induces the abscopal effect
- PMID: 40588370
- PMCID: PMC12211843
- DOI: 10.1136/jitc-2025-011487
Low-dose radiotherapy enhances the efficacy of PD-L1 blockade and induces the abscopal effect
Abstract
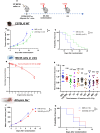
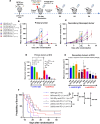
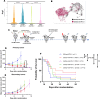
Background Low-dose radiotherapy (RT) is a promising treatment likely to increase the efficacy of immunotherapy, including programmed cell death ligand 1 (PD-L1) blockade, in cancer therapy. Further exploration and optimization of such combinatorial strategies are required. Notably, the ability of low-dose RT to enhance the efficacy of immune-checkpoint inhibitors (ICI) in distant, unirradiated tumors is debated. Methods We used a stepwise preclinical approach in immunocompetent mice bearing different murine tumor models (MC38 or CT26), with one or two tumors per mouse. Mice received tumor-only irradiation consisting of either low-dose RT (2x0.5 Gy to 2x2 Gy) or high-dose RT (2x6 Gy to 2x8 Gy) combined with anti-PD-L1. Tumor growth rate and survival were compared across the different conditions. The immune microenvironments of both irradiated and distant unirradiated tumors were characterized using single-cell RNA sequencing. Results We first demonstrated that low-dose RT 2×2 Gy combined with anti-PD-L1 is as effective as high-dose RT 2×6 Gy in delaying the growth of irradiated tumors. Subsequently, we showed that low-dose RT to one tumor enhances the efficacy of anti-PD-L1 consolidation therapy in a distant, unirradiated tumor, thereby inducing an abscopal effect comparable to that observed with high-dose RT. Single-cell RNA sequencing analysis highlighted the polarization of tumor-associated macrophages (TAMs) within distant unirradiated tumors towards a pro-inflammatory phenotype following low-dose RT and anti-PD-L1. Depleting TAMs in distant unirradiated tumors using liposomal clodronate abrogated the abscopal effect driven by low-dose RT combined with anti-PD-L1. Conclusion Our findings demonstrate the ability of low-dose RT to increase the efficacy of ICI in a distant tumor, resulting in a significant abscopal effect, and highlight the critical role of TAMs in the underlying mechanism, as well as a potential immune crosstalk between TAMs and activated lymphoid cells. These data propose low-dose RT as a potential strategy to improve the efficacy of immunotherapy in patients with metastatic solid tumors receiving anti-PD-L1.
Keywords: Abscopal; Immunotherapy; Macrophages; Radiotherapy/radioimmunotherapy; Solid tumor.
© Author(s) (or their employer(s)) 2025. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ Group.
Conflict of interest statement
Competing interests: ED reports grants and personal fees from Roche Genentech; grants from Servier; grants from AstraZeneca; grants and personal fees from MerckSerono; grants from BMS; and grants from MSD outside the submitted work. The other authors have declared that no conflict of interest exists.
Figures




References
-
- André T, Lonardi S, Wong KYM, et al. Nivolumab plus low-dose ipilimumab in previously treated patients with microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer: 4-year follow-up from CheckMate 142. Ann Oncol. 2022;33:1052–60. doi: 10.1016/j.annonc.2022.06.008. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
