Phase II basket trial of Dual Anti-CTLA-4 and anti-PD-1 blockade in Rare Tumors (DART) SWOG S1609: pancreatic neuroendocrine neoplasm (PNEN) cohort
- PMID: 40588371
- PMCID: PMC12211841
- DOI: 10.1136/jitc-2025-011760
Phase II basket trial of Dual Anti-CTLA-4 and anti-PD-1 blockade in Rare Tumors (DART) SWOG S1609: pancreatic neuroendocrine neoplasm (PNEN) cohort
Abstract
Purpose: SWOG S1609 Dual Anti-CTLA-4 and anti-PD-1 blockade in Rare Tumors (DART) studied the efficacy of ipilimumab combined with nivolumab across multiple rare tumor types. We report the results of the pancreatic neuroendocrine neoplasm (PNEN) cohort.
Experimental design: Treatment consisted of ipilimumab 1 mg/kg intravenously every 6 weeks with nivolumab 240 mg intravenously every 2 weeks. The primary endpoint was overall response rate (ORR) (Response Evaluation Criteria In Solid TumorsRECIST V.1.1). Secondary endpoints include progression-free survival (PFS), overall survival (OS), and toxicity. Clinical benefit rate (includes ORR plus stable disease (SD)>6 months was examined. Correlative studies were performed. The trial was conducted by the National Cancer Institute/Southwest Oncology Group Early Therapeutics and Rare Cancers Committee and opened at >1,000 sites.
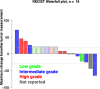
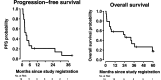
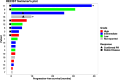
Results: 19 patients with PNEN were enrolled. The median number of lines of prior therapy was 2 (range: 0-4). The ORR was 11% (2/19 patients); the clinical benefit rate (CBR; stable disease >6 months+partial response+complete response), 26% (5/19). The median PFS was 3 months; median OS, 24 months. The longest PFSs were 26 (intermediate grade PNEN), 31 (low grade) and 39+months (intermediate grade). The most common toxicities were fatigue (47% of patients) and aspartate aminotransferase (AST) elevation (32%); the most common grade 3/4 immune-related adverse event (AE) was AST (32%) and bilirubin elevation (26%), with no grade 5 events. Programmed death-ligand 1 expression by chromogenic immunohistochemistry (N=12 patients assessed) did not associate with ORR; tumor mutation burden (TMB) was high in three patients; one of the two patients with partial remission (PFS=26 months) had high TMB (150 mutations/mb). Peripheral effector memory T-cell activation (N=11 patients assessed by cytometry by time-of-flight with 5 having longitudinal analysis) was associated with response, though the number of patients evaluated was limited.
Conclusions: Low-dose ipilimumab plus nivolumab demonstrated an 11% ORR and 26% CBR (includes SD>6 months) in patients with refractory PNEN, with durable benefit (>2 years) in 3 (16%) patients.
Trial registration number: NCT02834013.
Keywords: Immune Checkpoint Inhibitor; Ipilimumab; Neuroendocrine and Adrenal Tumor; Nivolumab; Solid tumor.
© Author(s) (or their employer(s)) 2025. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ Group.
Conflict of interest statement
Competing interests: RK has received research funding from Boehringer Ingelheim, Debiopharm, Foundation Medicine, Genentech, Grifols, Guardant, Incyte, Konica Minolta, Medimmune, Merck Serono, Omniseq, Pfizer, Sequenom, Takeda, and TopAlliance and from the NCI; as well as consultant and/or speaker fees and/or advisory board/consultant for Actuate Therapeutics, AstraZeneca, Bicara Therapeutics, Inc., Biological Dynamics, Caris, Datar Cancer Genetics, Daiichi, EISAI, EOM Pharmaceuticals, Iylon, LabCorp, Merck, NeoGenomics, Neomed, Pfizer, Precirix, Prosperdtx, Regeneron, Roche, TD2/Volastra, Turning Point Therapeutics, X-Biotech; has an equity interest in CureMatch Inc.; serves on the Board of CureMatch and CureMetrix, and is a co-founder of CureMatch. CWR received grants through his institution from Ayala, Bristol-Meyer Squibb, Daiichi-Sankyo, Deciphera, Exelixis, Genentech, Novartis, Karyopharm Therapeutics, Merck, Nektar, Pfizer, Xynomic, PF Argentum IP Holdings LLC, Rain Therapeutics, Shasqi, PTC Therapeutics, NiKang Therapeutics; consulting fees from Synox, Daiichi Sankyo, AVEO, Exelixis, Astra Zeneca, Bristol-Meyer Squibb; payment for expert testimony from Pfizer, GSK, Boehringer Ingelheim. JJL received partial funding from CA016672 from National Cancer Institute. IIW received consulting fees from Caris Life Sciences - Virtual molecular tumor board; payments from Advisory boards for AstraZeneca, Daiichi Sankyo, Takeda, Novartis, EMD Serono, Janssen, Pfizer, Eli Lilly and Company, Bayer, Regeneron, BMS and Genentech. Payment as Speaker from Blueprint Medicines, Janssen, Mirati and Takeda; support for attending meetings/travel from AnHeart Therapeutics; stock or stock options from MBrace Therapeutics. SP received grants or contracts from Amgen, AstraZeneca, A2bio, Bristol-Myers Squibb, Eli Lilly, Fate Therapeutics, Gilead, Iovance, Merck, Pfizer, Roche/Genentech; consulting fees from Amgen, AstraZeneca, BeiGene, Bristol-Myers Squibb, Eli Lilly, Genentech, Merck, Pfizer, Zai Labs. ES received consulting fees from D.E. Shaw Research; participated on a Data Safety Monitoring Board or Advisory Board for Mallinckrodt Pharmaceuticals. AK received grants or contracts through institution from TESARO, Astellas Pharma, Rafael Pharmaceuticals, Geistlich Pharma, Cardiff Oncology, FibroGen, Bavarian Nordic, Novocure, Cend Therapeutics, Ability Pharma, Novita, Boundless Bio; consulting fees from Ipsen; Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational eventsfrom Cardinal Health; Participation on a Data Safety Monitoring Board or Advisory Board for Ipsen. CLH received grants or contracts through institution from Sanofi, Avenge, Iovance, KSQ, Dragonfly, BTG, Novartis, 280Bio/Yingli, 4D Pharma, Medimmune/AZ, EMD Serono/Merck, Takeda, Obsidian, Genentech, BMS, Summit Therapeutics, Artidis, Immunogenesis; consulting fees from Regeneron; support for travel from Society for Immunotherapy of Cancer; Patents planned, issued or pending 62/977, 672 Managed by institution; stock or stock options from Briacell. DYD reports support from NIH CIMAC grant through institution; speaker honorarium from One Lambda. BK reported grant or contract through institution from Merck, Eisai, Xencor (ended in 8/2022). SG reports grant or contract from Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Genentech, Regeneron, and Takeda (Not as PI, payment to Institution, unrelated); consulting fee from Taiho Pharmaceuticals. MO reports support from NIH/NCI grant U10CA180819. HTM reports support from NIAID through institution and from Bill & Melinda Gates Foundation through institution; royalties or licenses from Standard BioTools; support for attending meetings/travel from Alamar Biosciences, Curiox Biosciences; Profiling and Treatment of MYC-Associated Cancers; Patents planned, issued or pending: U.S. Patent 11,648,275; SITC Biomarkers Committee - Leadership or fiduciary role in other board, society, committee or advocacy group, paid or unpaid; stock or stock options from BD Biosciences Cytek, Inc. JZ reports support from National Cancer Institute of the National Institute of Health Research Project Grant (R01CA234629), the AACR-Johnson & Johnson Lung Cancer Innovation Science Grant (18-90-52-ZHAN), the MD Anderson Physician Scientist Program, MD Anderson Lung Cancer Moon Shot Program; consulting fees from Johnson and Johnson, AstraZeneca, Novartis; Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Novartis, Bristol Myers Squibb, AstraZeneca, GenePlus, Innovent and Hengrui, St. Lucia, BeiGene; Participation on a Data Safety Monitoring Board or Advisory Board for Novartis, AstraZeneca, GenePlus, Catalyst, Helius, Oncohost, St Lucia; Receipt of equipment, materials, drugs, medical writing, gifts or other services from Novartis, Johnson and Johnson, Merck, Summit, Helius.
Figures

References
-
- Wagner MJ, Othus M, Patel SP, et al. Multicenter phase II trial (SWOG S1609, cohort 51) of ipilimumab and nivolumab in metastatic or unresectable angiosarcoma: a substudy of dual anti-CTLA-4 and anti-PD-1 blockade in rare tumors (DART) J Immunother Cancer. 2021;9:e002990. doi: 10.1136/jitc-2021-002990. - DOI - PMC - PubMed
-
- Adams S, Othus M, Patel SP, et al. A Multicenter Phase II Trial of Ipilimumab and Nivolumab in Unresectable or Metastatic Metaplastic Breast Cancer: Cohort 36 of Dual Anti-CTLA-4 and Anti-PD-1 Blockade in Rare Tumors (DART, SWOG S1609) Clin Cancer Res. 2022;28:271–8. doi: 10.1158/1078-0432.CCR-21-2182. - DOI - PMC - PubMed
-
- Chae YK, Othus M, Patel S, et al. A phase II basket trial of dual anti-CTLA-4 and anti-PD-1 blockade in rare tumors (DART) SWOG S1609: the thyroid tumor cohort. J Immunother Cancer. 2020;8:A161–A61. doi: 10.1136/jitc-2020-SITC2020.0270. - DOI
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- UG1 CA233331/CA/NCI NIH HHS/United States
- P30 CA016672/CA/NCI NIH HHS/United States
- UG1 CA233320/CA/NCI NIH HHS/United States
- U24 CA224319/CA/NCI NIH HHS/United States
- R33 CA263705/CA/NCI NIH HHS/United States
- U01 DK124165/DK/NIDDK NIH HHS/United States
- U10 CA180821/CA/NCI NIH HHS/United States
- U24 CA224316/CA/NCI NIH HHS/United States
- U24 CA224285/CA/NCI NIH HHS/United States
- U10 CA180828/CA/NCI NIH HHS/United States
- UG1 CA233198/CA/NCI NIH HHS/United States
- U10 CA180819/CA/NCI NIH HHS/United States
- U24 CA224309/CA/NCI NIH HHS/United States
- U10 CA180888/CA/NCI NIH HHS/United States
- P30 CA196521/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
