Inhibition of Wnt/β-catenin increases anti-tumor activity by synergizing with sorafenib in hepatocellular carcinoma
- PMID: 40593458
- PMCID: PMC12216529
- DOI: 10.1038/s41419-025-07789-5
Inhibition of Wnt/β-catenin increases anti-tumor activity by synergizing with sorafenib in hepatocellular carcinoma
Abstract
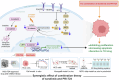
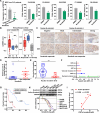
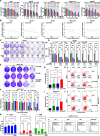
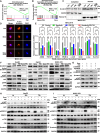
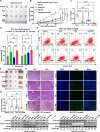
Hepatocellular carcinoma (HCC) poses a major global health challenge owing to limited treatment efficacy and drug resistance to therapies such as the tyrosine kinase inhibitor (TKI) sorafenib. We utilized a microfluidic three-dimensional (3D) drug testing system to assess drug responses in 37 fresh clinical samples and performed immunohistochemical analysis of 41 tumor tissue samples that received sorafenib therapy. Results revealed that Wnt/β-catenin activation was associated with sorafenib resistance, with higher nuclear β-catenin levels predicting poor response. Targeting Wnt/β-catenin via genetic intervention enhanced TKI sensitivity by promoting apoptosis and reducing clonogenicity. Through a large scale of drug and inhibitor library screening, we identified PRI-724, a potent CREB-binding protein (CBP)/β-catenin transcription antagonist, which synergistically induces apoptosis with sorafenib in vitro and in vivo by inhibiting β-catenin/CBP/c-myc, β-catenin nuclear localization and ERK/AKT signaling. The microfluidic 3D drug testing system confirmed the synergistic anti-tumor effects of this combination, underscoring its clinical application potential. Conclusively, our study provides a new combination therapy with sorafenib and PRI-724 to overcome TKI resistance and improve clinical outcomes in patients with HCC. Schematic representation of the speculative molecular mechanism model. Our study revealed that β-catenin activation drives sorafenib resistance in HCC, and disrupting β-catenin enhances sorafenib efficacy by promoting apoptosis and inhibiting proliferation. The combination of sorafenib and PRI-724, a Wnt/β-catenin inhibitor, showed synergistic anti-tumor effects in vitro across various HCC cell lines, in vivo using xenograft models, ex vivo utilizing MDT chip system to explore clinical applications, offering a novel therapeutic strategy for HCC patients.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests. Ethics approval and consent to participate: Informed consent was obtained from all participants involved in this study. Approval for this clinical research was granted by the Medical Ethics Committee of Tianjin Medical University Cancer Institute and Hospital (Approval number: bc2021260). And the study-related animal experiments were approved by the Experimental Animal Ethics Committee (Approval number: PMIF-2021077) of Tianjin Medical University Cancer Institute and Hospital, and the study was conducted in compliance with the Declaration of Helsinki.
Figures



References
MeSH terms
Substances
Grants and funding
- 82173208/National Natural Science Foundation of China (National Science Foundation of China)
- 82103672/National Natural Science Foundation of China (National Science Foundation of China)
- 82203243/National Natural Science Foundation of China (National Science Foundation of China)
- 82373365/National Natural Science Foundation of China (National Science Foundation of China)
- 24JCYBJC00700/Natural Science Foundation of Tianjin City (Natural Science Foundation of Tianjin)
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

