Efficacy and potential mechanisms of jatrorrhizine on MNNG-induced chronic atrophic gastritis in rats based on serological metabolomics and molecular docking
- PMID: 40594515
- PMCID: PMC12215369
- DOI: 10.1038/s41598-025-05502-2
Efficacy and potential mechanisms of jatrorrhizine on MNNG-induced chronic atrophic gastritis in rats based on serological metabolomics and molecular docking
Abstract
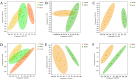
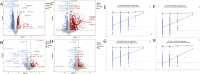
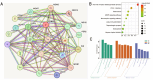
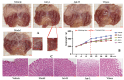
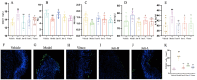
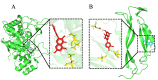
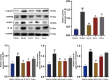
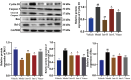
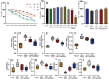
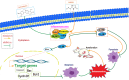
Jatrorrhizine (JATR), a natural isoquinoline alkaloid from Coptidis Rhizoma, exhibits various pharmacological activities, including antibacterial, anti-inflammatory, and antitumor effects. While JATR is known to treat chronic gastritis, its therapeutic potential for chronic atrophic gastritis (CAG) and its underlying mechanisms are not fully understood. This study induced CAG in rats using N-Methyl-N'-nitro-N-nitrosoguanidine (MNNG) for 12 weeks through free drinking and force-feeding. Serological metabolomics identified 23 core targets of JATR related to CAG improvement. Reverse transcription-quantitative polymerase chain reaction and western blotting confirmed the involvement of these targets. Molecular docking revealed interactions between JATR and IL-1β and Caspase-3. JATR significantly alleviated gastric inflammation and atrophy, with Kyoto Encyclopedia of Genes and Genomes analysis showing enrichment in the "Nod-like receptor-related pyroptosis pathway". JATR also enhanced GES-1 cell proliferation and reduced MNNG-induced cell damage. Additionally, JATR downregulated pyroptosis-related (Gasdermin D, NLRP3, Caspase-1) and apoptosis-related (Bcl-2, Bax, Caspase-3) markers. These findings suggest that JATR may ameliorate MNNG-induced CAG by inhibiting the activation of the Nod-like receptor-related pyroptosis pathway, supporting its potential as a therapeutic intervention for CAG.
Keywords: Caspase-3; Chronic atrophic gastritis; Jatrorrhizine; MNNG; Molecular docking; NL family pyrin domain containing 3.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Ethics statement: All animal procedures were carried out by following the Guide for the Care and Use of Laboratory. The research was approved by the Chongqing Academy of Animal Science Animal Ethics Committee (Approval ID: XKY-20240820). This study is reported following ARRIVE guidelines (Animal Research: Reporting of In Vivo Experiments).
Figures


Similar articles
-
Jatrorrhizine attenuates inflammatory response in Helicobacter pylori-induced gastritis by suppressing NLRP3 inflammasomes and NF-κB signaling pathway.Arch Microbiol. 2025 Jun 17;207(8):177. doi: 10.1007/s00203-025-04374-4. Arch Microbiol. 2025. PMID: 40526168
-
Zuojin Pill attenuates Helicobacter pylori-induced chronic atrophic gastritis in rats and improves gastric epithelial cells function in GES-1 cells.J Ethnopharmacol. 2022 Mar 1;285:114855. doi: 10.1016/j.jep.2021.114855. Epub 2021 Nov 20. J Ethnopharmacol. 2022. PMID: 34808298
-
Formononetin Alleviates MNNG-Triggered Chronic Atrophic Gastritis: Its Potential Mechanisms.Comb Chem High Throughput Screen. 2025 Jul 3. doi: 10.2174/0113862073409559250618212035. Online ahead of print. Comb Chem High Throughput Screen. 2025. PMID: 40619665
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
References
-
- Tan, P. & Yeoh, K. G. Genetics and molecular pathogenesis of gastric adenocarcinoma. Gastroenterology149, 1153-1162.e1153. 10.1053/j.gastro.2015.05.059 (2015). - PubMed
-
- Graham, D. Y. & Fischbach, L. Helicobacter pylori treatment in the era of increasing antibiotic resistance. Gut59, 1143–1153. 10.1136/gut.2009.192757 (2010). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

