Biotransformation of medicarpin from homopterocarpin by Aspergillus niger and its biological characterization
- PMID: 40596118
- PMCID: PMC12215371
- DOI: 10.1038/s41598-025-06729-9
Biotransformation of medicarpin from homopterocarpin by Aspergillus niger and its biological characterization
Erratum in
-
Correction: Biotransformation of medicarpin from homopterocarpin by Aspergillus niger and its biological characterization.Sci Rep. 2025 Jul 11;15(1):25056. doi: 10.1038/s41598-025-11009-7. Sci Rep. 2025. PMID: 40646145 Free PMC article. No abstract available.
Abstract
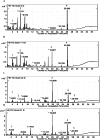
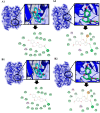
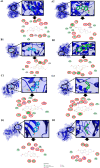
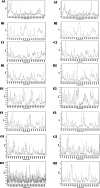
The objective of this study was to convert homopterocarpin derived from Pterocarpus macrocarpus Kurz. heartwood to medicarpin using Aspergillus niger (strain UI X-172) and assess its antioxidant, antiplasmodial, and anticancer activities in silico and in vitro. This study highlighted biotransformation of homopterocarpin to medicarpin via demethylation. Medicarpin demonstrated antioxidant activity against 2,2-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS, IC50 = 0.61 ± 0.05 µg/mL) and 1,1-diphenyl-2-picrylhydrazyl (DPPH, IC50 = 7.50 ± 1.6 µg/mL), antiplasmodial activity against the Plasmodium falciparum strain 3D7 (IC50 = 0.45 ± 0.35 µg/mL), and anticancer efficacy against a hepatocyte-derived carcinoma cell line (Huh7it-1 cells, IC50 = 34.32 ± 5.56 µg/mL). Medicarpin also showed favorable antioxidant, antiplasmodial, and anticancer properties in silico with a binding affinity lower than commercial drugs. These results highlight the green synthesis of medicarpin by microbial transformation using A. niger, which demonstrates promising in vitro and computational activity, however, further studies are required for clinical development.
Keywords: Aspergillus Niger; Biological properties; Biotransformation; Homopterocarpin; Medicarpin.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures



References
-
- Anwar, Y., Putra, A. M. J., Iftitah, E. D., Simanjuntak, P. & Kumala, S. Prediction of geraniol bond mode in Aspergillus Niger Linalool Dehydratase – Isomerase. Maj Obat Tradis. 24, 133 (2019).
-
- Chatla, S., Gasi, S., Rahaman, S. A., Yaradla, M. & Mareedu, H. Biotransformation of quercetin by Aspergillus Niger from Allium cepa. IJPSR12, 3210–3215 (2021).
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

