Circulating tumor cells and clusters as liquid biomarkers for the diagnosis and prognosis of neuroblastoma
- PMID: 40597797
- PMCID: PMC12211310
- DOI: 10.1186/s12885-025-14448-7
Circulating tumor cells and clusters as liquid biomarkers for the diagnosis and prognosis of neuroblastoma
Abstract
Background: The clinical significance of circulating tumor cell (CTC) clusters in highly metastatic tumors hasn't yet been revealed. Here, we demonstrated the diagnostic and prognostic value of CTC clusters in neuroblastoma (NB) which is the most prevalent childhood extracranial solid tumor.
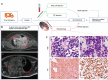
Methods: We employed cascaded filter deterministic lateral displacement microfluidic chips to enrich CTCs and CTC clusters in 64 newly diagnosed NB patients. CTCs and CTC clusters were identified by CD45-, GD2+/PHOX2B+, DAPI + immunofluorescence staining, with cells displaying characteristic neoplastic morphology.
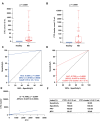
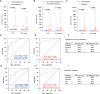
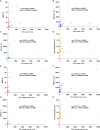
Results: Among NB patients, 85.94% and 50.00% were positive for CTCs and CTC clusters, respectively; no CTCs or CTC clusters were detected in healthy children. Moreover, CTC and CTC cluster numbers differed significantly across different primary sites, clinical and pathologic features, and risk stratifications, while no significant differences in CTC and CTC cluster counts were observed in relation to sex, age, and MYCN gene amplification. CTCs and CTC clusters indicated metastasis and strongly correlated with minimal residual disease. Of note, CTC clusters ≥ 2.5/2 mL were closely associated with bone marrow metastasis and demonstrated significant differences in the hazard ratio of overall survival.
Conclusions: CTCs and CTC clusters are sensitive non-invasive biomarkers for NB diagnosis and prognosis, especially the prominent role in tumor emergencies. CTC clusters closely correlate with bone marrow metastasis and represent promising indicator for the monitoring of metastasis in NB emergencies. The mechanisms of CTC cluster formation and their specific role in the metastasis cascade deserve further elucidation which may serve as targets to inhibit NB bone marrow metastasis.
Keywords: Circulating tumor cell; Circulating tumor cell cluster; Liquid biopsy; Microfluidic chip; Neuroblastoma.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Ethical approval for the research was granted from the Shenzhen Children’s Hospital Ethics Committee. All patients consented to donate samples to this study. The study was performed in accordance with the Declaration of Helsinki. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures



References
MeSH terms
Substances
Grants and funding
- SZSM 202211033/Sanming Project of Medicine in Shenzhen
- SZSM 202211033/Sanming Project of Medicine in Shenzhen
- SZSM 202211033/Sanming Project of Medicine in Shenzhen
- SZSM 202211033/Sanming Project of Medicine in Shenzhen
- SZSM 202211033/Sanming Project of Medicine in Shenzhen
- SZSM 202211033/Sanming Project of Medicine in Shenzhen
- SZSM 202211033/Sanming Project of Medicine in Shenzhen
- SZSM 202211033/Sanming Project of Medicine in Shenzhen
- SZSM 202211033/Sanming Project of Medicine in Shenzhen
- SZGSP012/Shenzhen Fund for Guangdong Provincial High-level Clinical Key Specialties
- SZGSP012/Shenzhen Fund for Guangdong Provincial High-level Clinical Key Specialties
- SZGSP012/Shenzhen Fund for Guangdong Provincial High-level Clinical Key Specialties
- SZGSP012/Shenzhen Fund for Guangdong Provincial High-level Clinical Key Specialties
- SZGSP012/Shenzhen Fund for Guangdong Provincial High-level Clinical Key Specialties
- SZGSP012/Shenzhen Fund for Guangdong Provincial High-level Clinical Key Specialties
- SZGSP012/Shenzhen Fund for Guangdong Provincial High-level Clinical Key Specialties
- SZGSP012/Shenzhen Fund for Guangdong Provincial High-level Clinical Key Specialties
- SZGSP012/Shenzhen Fund for Guangdong Provincial High-level Clinical Key Specialties
- szcrc2024_003/Shenzhen Clinical Research Center for Child Health and Disease
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

