Effects of pulse glucocorticoid treatment on retinal blood flow in patients with active Graves' orbitopathy
- PMID: 40597947
- PMCID: PMC12220388
- DOI: 10.1186/s12886-025-04184-8
Effects of pulse glucocorticoid treatment on retinal blood flow in patients with active Graves' orbitopathy
Abstract
Background: Various studies have shown decreased retinal blood flow in patients with active Graves' orbitopathy (GO). We investigated whether retinal blood flow returned to normal levels after intravenous glucocorticoid treatment in active GO patients.
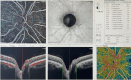
Methods: Thirty-two eyes of 32 patients with moderate or severe active GO and 32 eyes of 32 healthy participants were included. The blood flow in the superficial plexus (SP), deep retinal plexus (DP), external retinal (ER) capillary plexus and choriocapillaris was measured via OCT-Angiography before treatment with methylprednisolone and one week after the completion of treatment. In addition, central macular thickness, intraocular pressure, and best corrected visual acuity, were compared with those of the control group.
Results: Retinal blood flow (RBF) in the macula at SP, DP, ER and choriocapillaris levels was 48.38 ± 2.04, 54.17 ± 3.11, 0.42 ± 0.14 and 2176.45 ± 147.53 in the control group and 45.91 ± 4.56, 51.55 ± 3.84, 0.35 ± 0.34 and 2030.15 ± 132.15 in the active GO group, respectively. The RBF was significantly lower in the active GO group than in the control group at all angiographic levels (p < 0.001). Although blood flow increased after treatment, it remained lower than that in the control group (p < 0.001). The mean CMT was significantly greater in active GO patients before treatment than in the controls and after treatment (p < 0.001). No serious systemic or ocular side effects were observed in any of the patients.
Conclusions: After pulse glucocorticoid treatment, retinal blood flow increases but remains low compared with that in the healthy control group.
Keywords: Glucocorticoids; Graves’ ophthalmopathy; Optical coherence tomography angiography; Pulse therapy; Vessel, retinal blood.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval: For the present study, approval was obtained from the Local Ethics Committee for Clinical Practices of Gaziantep University (Reg. number: 07/25/2021 − 205) under the 1961 Declaration of Helsinki and its later amendments. Informed consent was obtained from all individual participants included in this study. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures
References
-
- Furmaniak J, Sanders J, Sanders P, et al. TSH receptor-specific monoclonal autoantibody K1-70TM targeting of the TSH receptor in subjects with graves’ disease and graves’ orbitopathy-Results from a phase I clinical trial. Clin Endocrinol (Oxf). 2022;96(6):878–87. 10.1111/cen.14681. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical