Aurantio-obtusin modulates Wilms Tumour 1 within the breast tumour microenvironment reducing immunosuppression and tumour growth
- PMID: 40598415
- PMCID: PMC12211983
- DOI: 10.1186/s12964-025-02292-y
Aurantio-obtusin modulates Wilms Tumour 1 within the breast tumour microenvironment reducing immunosuppression and tumour growth
Abstract
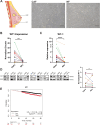
Introduction: Cancer associated fibroblasts (CAFs) contribute to tumourigenesis and immune tolerance within the tumour microenvironment (TME). Therefore, inhibiting the pro-tumourigenic function of CAFs can be a viable therapeutic approach. However, targeting CAFs is challenging due to the lack of specific markers. The objective of this study is to identify CAF specific therapeutic targets that have the potential to enhance tumour immunity and reduce tumour growth.
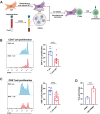
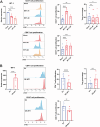
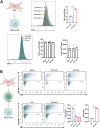
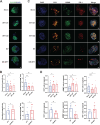
Methods: RNA sequencing was performed on CAFs and normal fibroblasts (NFs) from the same breast cancer patient. Wilms tumour-1 (WT1) was identified as a gene upregulated in CAFs. WT1 levels in CAFs were manipulated using plasmid overexpression of-or siRNA downregulation of WT1. Co-culture assays were performed to evaluate the role of CAF-derived WT1 in T cell proliferation and differentiation using flow cytometry. Western blot and ELISA were performed to interrogate the mechanism of action of WT1 within CAFs. Three-dimensional patient-derived organoids (PDOs) that encompassed the tumour immune-microenvironment were established to determine the therapeutic potential of targeting CAF-derived WT1.
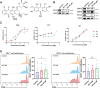
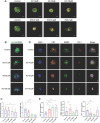
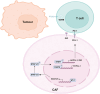
Results: WT1, a transcription factor, regulates signal transducer and activator of transcription (STAT) 1/3 levels, promotes programmed death ligand 1 (PD-L1) expression and indoleamine 2,3-dioxygenase (IDO) expression in CAFs. CAF-derived WT1 reduces the proliferation of CD4+ and CD8+ T cells and enhances the differentiation of naïve T cells into regulatory T cells (Tregs), thus producing an immunosuppressive TME. Reducing CAF WT1 levels results in less immunosuppressive CAFs, smaller PDOs and increased levels of cytotoxic granzyme B+ (GZMB+) T cells within the TME. Standard chemotherapeutic agents, paclitaxel (PTX) and doxorubicin (DOX), increase WT1 levels in CAFs enhancing their ability to suppress T cell proliferation. However, Aurantio-obtusin (AO, a DOX analogue) decreases WT1 expression in CAFs reducing their ability to suppress T cell proliferation. AO causes decreased PDO size which correlates with increased levels of T cells within the TME.
Conclusions: Therapeutic targeting of the WT1/STAT1/3/PD-L1/IDO axis in CAFs with AO has the potential to enhance T cell activity and reduce Treg percentage within the TME, thereby enhancing tumour immunity and reducing tumourigenesis.
Keywords: Breast cancer; Cancer associated fibroblast; Immunosuppression; Tumour microenvironment; Wilms’ tumour 1.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Ethical approval was obtained from the research ethics committee at the University of Galway (Ref: 45/05). After written informed consent, fresh specimens of human breast tumours were harvested from patients undergoing surgery at University College Hospital Galway. All patient material and clinical information were obtained from the Cancer Biobank at the University of Galway. Consent for publication: Not applicable. Competing interests: SJE and PGL are employees and shareholders of Orbsen Therapeutics Ltd. RL, DOC, BD, POB, XH, NG, MK and LRB have no conflicts of interest to declare.
Figures


Similar articles
-
Cancer-associated fibroblast-derived extracellular vesicles loaded with GLUT1 inhibitor synergize anti-PD-L1 to suppress tumor growth via degrading matrix stiffness and remodeling tumor microenvironment.J Control Release. 2025 Sep 10;385:113998. doi: 10.1016/j.jconrel.2025.113998. Epub 2025 Jul 1. J Control Release. 2025. PMID: 40609836
-
Ectopic expression of GDF15 in cancer-associated fibroblasts enhances melanoma immunosuppression via the GFRAL/RET cascade.J Immunother Cancer. 2025 Jun 24;13(6):e011036. doi: 10.1136/jitc-2024-011036. J Immunother Cancer. 2025. PMID: 40555562 Free PMC article.
-
Constructing a co-culture model of cancer-associated fibroblasts and ovarian cancer organoids and studying mechanisms of drug resistance.Exp Cell Res. 2025 Jul 15;450(2):114656. doi: 10.1016/j.yexcr.2025.114656. Epub 2025 Jun 23. Exp Cell Res. 2025. PMID: 40562199
-
Cost-effectiveness of using prognostic information to select women with breast cancer for adjuvant systemic therapy.Health Technol Assess. 2006 Sep;10(34):iii-iv, ix-xi, 1-204. doi: 10.3310/hta10340. Health Technol Assess. 2006. PMID: 16959170
-
Targeting the devil: Strategies against cancer-associated fibroblasts in colorectal cancer.Transl Res. 2024 Aug;270:81-93. doi: 10.1016/j.trsl.2024.04.003. Epub 2024 Apr 16. Transl Res. 2024. PMID: 38614213 Review.
References
-
- Wang R, Zeng H, Xiao X, Zheng J, Ke N, Xie W, Lin Q, Zhang H. Identification of prognostic biomarkers of breast cancer based on the immune-related gene module. Autoimmunity. 2023;56(1):2244695. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

