Experiences, impact, and enablers of involving young people and family caregivers in developing reporting guidelines for paediatric randomised trials: a case study
- PMID: 40598557
- PMCID: PMC12211127
- DOI: 10.1186/s40900-025-00751-x
Experiences, impact, and enablers of involving young people and family caregivers in developing reporting guidelines for paediatric randomised trials: a case study
Abstract
Background: Patient and public involvement (PPI) is increasingly recognized as important, yet no guidance exists on integrating young people and family caregiver perspectives in the development of research reporting guidelines. We developed two paediatric-specific extensions with young people (ages 10-24 years) and family caregivers (YPFC) for the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) and Consolidated Standards of Reporting Trials (CONSORT) reporting guidelines: SPIRIT-Children and Adolescents 2025 (SPIRIT-C) and CONSORT-Children and Adolescents 2025 (CONSORT-C). This case study describes how we involved YPFC in the development of SPIRIT-C and CONSORT-C and identified enablers of impactful PPI.
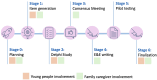
Main text: We formed a Youth Advisory Group (ages 13-19 years) and a Family Caregiver Advisory Group. A miniseries of two Young Person Reporting Guideline workshops aimed at generating randomised controlled trials (RCT) reporting items were conducted virtually in Canada, England, France, Scotland, and Spain, engaging 42 young people (ages 10-21 years). Young people (ages 19-24 years) and family caregivers participated as panellists in an international Delphi study. Family caregiver advisors actively contributed to the Consensus Meeting and to the writing process of the guidelines' Explanation and Elaboration (E&E) documents. After each project stage, YPFC feedback was collected. PPI impact was defined as tangible changes, learnings, and outcomes, both positive and negative, to the guideline development process and the final guidelines resulting from YPFC co-development. YPFC found their involvement in the project a valuable experience. Their contributions to key project stages, such as the Delphi study, Consensus Meeting, and the development of the E&E documents impacted the final guidelines and E&E documents, with the inclusion of four new youth generated reporting items. Feedback throughout the project informed six "enablers" for productive partnerships in reporting guideline development: (1) designated point person, (2) tailored training, (3) access to project materials, (4) clear expectations on time commitment and compensation, (5) structured check-in sessions, and (6) demonstrated openness to feedback.
Conclusion: With careful preparation, investing in impactful PPI enablers, YPFC can meaningfully contribute to the development of research reporting guidelines, improve final deliverables, and ultimately shape research that reflects their perspectives.
Keywords: Adolescents; Children; Clinical trial; Family involvement; Paediatrics; Patient and public involvement; Randomised controlled trial; Reporting guideline; Youth involvement.
Plain language summary
Young people and family caregivers (YPFC) are end-users of paediatric research reports. To ensure that items of importance to them are reported, they should have a say in what key items of a study should be reported. While there is increased interest in involving YPFC in health research, guidance on how best to involve young people ages 10–24 years and family caregivers in the development of research reporting guidelines does not exist. Recently, recommendations on how patient and public partners can be meaningfully involved in research reporting guideline development were published, though these recommendations were informed by the experiences of adult partners. This study is the first to involve YPFC in the development of reporting guidelines for paediatric randomised controlled trials (RCTs). It describes the experience of this involvement from multiple perspectives and evaluates YPFC’s impact on the final guidelines. YPFCs contributed to the generation of candidate reporting items, a three-round Delphi study, Consensus Meeting, and writing/review of the Explanation and Elaboration documents. Using formal evaluations after each project stage, we identified six enablers for their effective involvement in future projects that aim to add value to child health research by integrating YPFCs perspectives. Investing in enablers for effective YPFC involvement may improve research practice and yield increased relevance and utility of child health research deliverables and outcomes for end-users and add value to research reflecting YPFC perspectives.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The Research Ethics Board of The Hospital for Sick Children confirmed that ethics approval is not required as we worked young people and family caregivers in an advisory capacity. Consent for publication: Not applicable. Competing interests: KC declares grants/contracts from Ontario Child Health Support Unit to support projects that foster family engagement in research and was offered honoraria as a Family Caregiver Advisor of the SPIRIT | CONSORT-C project. ADK, LM, and TCN received honoraria for their role as a Family Caregiver Advisor of the SPIRIT | CONSORT-C project. MS received honoraria for her role as the Patient Engagement Lead in the SPIRIT | CONSORT-C Core Project Team. All other authors declare no competing interests.
Figures
Similar articles
-
Do peer reviewers comment on reporting items as instructed by the journal? A secondary analysis of two randomized trials.J Clin Epidemiol. 2025 Jul;183:111818. doi: 10.1016/j.jclinepi.2025.111818. Epub 2025 May 8. J Clin Epidemiol. 2025. PMID: 40348145
-
Home treatment for mental health problems: a systematic review.Health Technol Assess. 2001;5(15):1-139. doi: 10.3310/hta5150. Health Technol Assess. 2001. PMID: 11532236
-
How lived experiences of illness trajectories, burdens of treatment, and social inequalities shape service user and caregiver participation in health and social care: a theory-informed qualitative evidence synthesis.Health Soc Care Deliv Res. 2025 Jun;13(24):1-120. doi: 10.3310/HGTQ8159. Health Soc Care Deliv Res. 2025. PMID: 40548558
-
Consolidated standards of reporting trials (CONSORT) and the completeness of reporting of randomised controlled trials (RCTs) published in medical journals.Cochrane Database Syst Rev. 2012 Nov 14;11(11):MR000030. doi: 10.1002/14651858.MR000030.pub2. Cochrane Database Syst Rev. 2012. PMID: 23152285 Free PMC article.
-
Transition of care for adolescents from paediatric services to adult health services.Cochrane Database Syst Rev. 2016 Apr 29;4(4):CD009794. doi: 10.1002/14651858.CD009794.pub2. Cochrane Database Syst Rev. 2016. PMID: 27128768 Free PMC article.
References
-
- Series from the Lancet journals: Research: increasing value, reducing waste: The Lancet. 2014 [Available from: https://www.thelancet.com/series/research
-
- Hopewell S, Boutron I, Chan AW, Collins GS, de Beyer JA, Hrobjartsson A, et al. An update to SPIRIT and CONSORT reporting guidelines to enhance transparency in randomized trials. Nat Med. 2022;28(9):1740–3. - PubMed
-
- Li Q, Zhou Q, Florez ID, Mathew JL, Amer YS, Estill J et al. Reporting standards for child health research were few and poorly implemented. J Clin Epidemiol. 2023. - PubMed
-
- EQUATOR Network. Enhancing the QUAlity and Transparency Of health Research [Available from: http://www.equator-network.org/reporting-guidelines/
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources