Hypoxia preconditioned MSC exosomes attenuate high-altitude cerebral edema via the miR-125a-5p/RTEF-1 axis to protect vascular endothelial cells
- PMID: 40599343
- PMCID: PMC12212178
- DOI: 10.1016/j.bioactmat.2025.06.018
Hypoxia preconditioned MSC exosomes attenuate high-altitude cerebral edema via the miR-125a-5p/RTEF-1 axis to protect vascular endothelial cells
Abstract
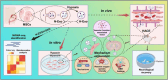
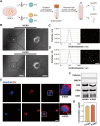
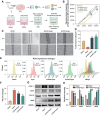
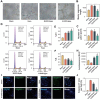
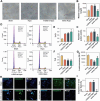
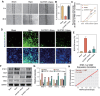
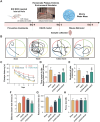
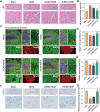
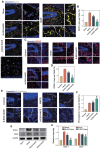
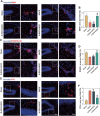
Vasogenic edema, caused by the disruption of the blood-brain barrier (BBB), is a significant pathological factor in high-altitude cerebral edema (HACE). Due to the rapid progression and high mortality rate of HACE, prophylactic treatment is important. Mesenchymal stem cell exosomes (MSC-EXO) are increasingly being used in tissue injury repair, and research suggests that appropriate conditioning can enhance the targeted efficacy of exosome therapy. Our in vitro experiments revealed that hypoxia preconditioned MSC-EXO (H-EXO) significantly outperformed normoxic MSC-EXO (N-EXO) in multiple protective aspects. Specifically, H-EXO demonstrated enhanced capacity to mitigate hypoxia-induced aberrant angiogenesis, maintain vascular endothelial cell viability, and suppress ROS accumulation and apoptotic signaling under hypoxic stress. Mechanistic investigation identified miR-125a-5p cargo in H-EXO as a key mediator of RTEF-1 targeted inhibition during hypoxic exposure. In corresponding in vivo studies, H-EXO administration effectively attenuated HACE-induced pathological angiogenesis while maintaining crucial vascular homeostasis markers. The therapeutic effects manifested through three principal aspects: 1) downregulation of RTEF-1/VEGF hyperexpression, 2) modulation of VE-cadherin, SMA, and PDGFRα + β expression to preserve BBB integrity, and 3) concurrent protection of neurovascular functions against HACE-induced damage. This investigation elucidates the miR-125a-5p/RTEF-1 axis as the central mechanism through which hypoxic preconditioning enhances MSC-EXO's endothelial protective properties. Our findings establish H-EXO's multimodal therapeutic potential, demonstrating its capacity to simultaneously inhibit pathological angiogenesis, restore BBB function, and protect neural tissue under hypoxic stress conditions. The study elucidates key mechanisms underlying clinical prevention and management of HACE by delineating H-EXO's preventive mechanisms against hypoxia-induced cerebrovascular injury.
Keywords: High-altitude cerebral edema; Hypoxia preconditioned exosomes; Neurological recovery; Vascular endothelial cells; miR-125a-5p.
© 2025 The Authors.
Conflict of interest statement
The authors declare the following personal relationships which may be considered as potential competing interests: Xiao-Li Zeng, ,Ling-Jie Zhang, Xin-Jian Yan and Ping Zhu are currently employed by Guangzhou Xiangxue Pharmaceutical Company limited, Guangdong Xiangxue Stem Cell Regenerative Medicine Technology Company limited.
Figures

References
-
- Hackett P.H., Roach R.C. High-altitude illness. N. Engl. J. Med. 2001;345(2):107–114. - PubMed
-
- Gatterer H., Villafuerte F.C., Ulrich S., Bhandari S.S., Keyes L.E., Burtscher M. Altitude illnesses. Nat. Rev. Dis. Primers. 2024;10(1):43. - PubMed
-
- Luks A.M., Hackett P.H. Medical conditions and high-altitude travel. N. Engl. J. Med. 2022;386(4):364–373. - PubMed
-
- Wu T.Y., Ding S.Q., Liu J.L., Jia J.H., Dai R.C., Liang B.Z., et al. Ataxia: an early indicator in high altitude cerebral edema. High Alt. Med. Biol. 2006;7(4):275–280. - PubMed
LinkOut - more resources
Full Text Sources

