GATA8-Mediated Antiviral Defence Is Countered by Tomato Chlorosis Virus-Encoded Pathogenicity Protein p27
- PMID: 40600259
- PMCID: PMC12214945
- DOI: 10.1111/mpp.70115
GATA8-Mediated Antiviral Defence Is Countered by Tomato Chlorosis Virus-Encoded Pathogenicity Protein p27
Abstract
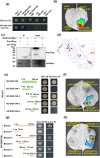
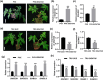
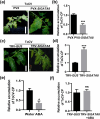
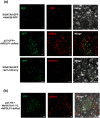
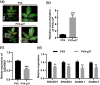
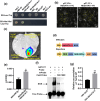
Tomato chlorosis virus (ToCV), a phloem-restricted RNA virus within the genus Crinivirus of the family Closteroviridae, exhibits a broad host range and severely impacts the yield and quality of multiple crops. Viral infection directly alters endogenous phytohormone levels, which are intricately associated with viral mobility, replication, symptom development and defence mechanisms. Previous studies have demonstrated that GATA transcription factors regulate several hormone signalling pathways in plants. In this study, we explored the interaction between ToCV p27 and SlGATA8/NbGATA11. Results indicated that ToCV p27 interacts with an 18-amino-acid at the C-terminus of SlGATA8 and NbGATA11 proteins. Silencing and overexpressing of SlGATA8 revealed its positive role in regulating tomato defence against ToCV infection. Additionally, the interaction redirected SlGATA8's subcellular localisation to plasmodesmata. Furthermore, SlGATA8 promoted the transcriptional expression of SlSnRK2 to regulate the abscisic acid (ABA) signalling pathway. In conclusion, this study confirmed that ToCV p27 impaired the transcriptional activation activity of SlGATA8 through direct interaction, thereby inhibiting the ABA pathway and ultimately facilitating viral infection. This study established a link among virus, GATA family transcription factors and phytohormones, elucidating the molecular mechanism by which ToCV-encoded p27 protein interacts with SlGATA8 to disrupt ABA balance and promote virus infection.
Keywords: GATA transcription factors; SlSnRK2; counterdefense; phytohormone; tomato chlorosis virus.
© 2025 The Author(s). Molecular Plant Pathology published by British Society for Plant Pathology and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Pathogenicity factor p22 of tomato chlorosis virus interferes with abscisic acid-mediated antiviral defenses by disrupting the function of WRKY81 in tomato.New Phytol. 2025 Aug;247(4):1837-1854. doi: 10.1111/nph.70267. Epub 2025 Jun 22. New Phytol. 2025. PMID: 40545789
-
Cathepsin F alters viral acquisition, retention, and transmission of TYLCV and ToCV by Bemisia tabaci MED.J Econ Entomol. 2025 Apr 26;118(2):486-494. doi: 10.1093/jee/toae269. J Econ Entomol. 2025. PMID: 39937204
-
Down-regulation of 14-3-3 genes suppress sugar metabolism in Bemisia tabaci MED and reduce transmission of tomato chlorosis virus.Pest Manag Sci. 2025 Sep;81(9):5763-5773. doi: 10.1002/ps.8929. Epub 2025 Jul 3. Pest Manag Sci. 2025. PMID: 40605785
-
Tomato brown rugose fruit virus: An emerging and rapidly spreading plant RNA virus that threatens tomato production worldwide.Mol Plant Pathol. 2022 Sep;23(9):1262-1277. doi: 10.1111/mpp.13229. Epub 2022 May 22. Mol Plant Pathol. 2022. PMID: 35598295 Free PMC article. Review.
-
Natural Resources Resistance to Tomato Spotted Wilt Virus (TSWV) in Tomato (Solanum lycopersicum).Int J Mol Sci. 2021 Oct 12;22(20):10978. doi: 10.3390/ijms222010978. Int J Mol Sci. 2021. PMID: 34681638 Free PMC article. Review.
References
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources

