The Efficacy and Safety of Single Injections of Hybrid HA in the Treatment of Symptomatic Knee Osteoarthritis: A Case Series
- PMID: 40601162
- PMCID: PMC12246308
- DOI: 10.1007/s40744-025-00780-8
The Efficacy and Safety of Single Injections of Hybrid HA in the Treatment of Symptomatic Knee Osteoarthritis: A Case Series
Abstract
Introduction: The prevalence of knee osteoarthritis (OA) is rising worldwide, leading to disability and a reduced quality of life, particularly in elderly patients. While there are several treatment options, there is little consensus in the scientific community over which methods are most effective. Viscosupplementation with hyaluronic acid (HA) has been found to reduce pain in patients with knee OA over a period of up to 6 months, with little to no side effects. The aim of this prospective open-label, uncontrolled, observational, single-site study was to assess the efficacy and safety of a single hybrid HA injection over a period of 6 months in subpopulations of patients with low to severe symptomatic knee OA in everyday clinical practice.
Methods: Fifty patients who met the inclusion criteria participated in the study. A single intra-articular ultrasound-guided injection of hybrid HA (Sinovial®) was administered. Patients submitted Visual Analog Scale (VAS) and Western Ontario and McMaster Universities Arthritis Index (WOMAC) questionnaires at 28, 42, 84, and 168 days post-treatment.
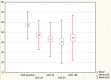
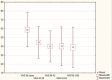
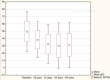
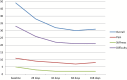
Results: VAS scores measured at rest and when walking indicate an improvement during follow-up, particularly at 28 and 42 days, compared to baseline. Similarly, the most notable improvement of the WOMAC score was observed within the first 42 days after injection. While decrease in pain and joint function improvement were not as pronounced at the end of follow-up, they were still statistically better than at baseline. Overall patient satisfaction was high.
Conclusion: Treatment with a single injection of hybrid HA was demonstrated to be safe and effective in patients with varying degrees of knee OA. Patients with medial knee OA responded better to treatment than patients with patellofemoral OA, which provides information on which types of patients are best suited to this intervention.
Trial registration: ClinicalTrials.gov identifier, NCT06652893. Retrospectively registered October 10, 2024.
Keywords: Hybrid HA; Osteoarthritis; VAS; Viscosupplementation; WOMAC.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of Interest: Marcin E. Domzalski and Klaudia Marchewa declare that they have no competing interests. Ethical Approval: The study was performed in accordance with the ethical guidelines of the Declaration of Helsinki and was approved by the Local Bioethics Committee in Lodz, Poland. Collected information and data were managed according to the guidelines of Good Clinical Practice (GCP). All participants signed an informed consent form at the time of enrollment to collect clinical data. All patients also gave their consent to the use of patient reported outcomes (PROs). The study was registered on ClinicalTrials.gov with NCT06652893.
Figures
Similar articles
-
Hyaluronic acid and other conservative treatment options for osteoarthritis of the ankle.Cochrane Database Syst Rev. 2015 Oct 17;2015(10):CD010643. doi: 10.1002/14651858.CD010643.pub2. Cochrane Database Syst Rev. 2015. PMID: 26475434 Free PMC article.
-
Intra-articular corticosteroid for knee osteoarthritis.Cochrane Database Syst Rev. 2015 Oct 22;2015(10):CD005328. doi: 10.1002/14651858.CD005328.pub3. Cochrane Database Syst Rev. 2015. PMID: 26490760 Free PMC article.
-
Celecoxib for osteoarthritis.Cochrane Database Syst Rev. 2017 May 22;5(5):CD009865. doi: 10.1002/14651858.CD009865.pub2. Cochrane Database Syst Rev. 2017. PMID: 28530031 Free PMC article.
-
Intraarticular corticosteroid for treatment of osteoarthritis of the knee.Cochrane Database Syst Rev. 2006 Apr 19;(2):CD005328. doi: 10.1002/14651858.CD005328.pub2. Cochrane Database Syst Rev. 2006. Update in: Cochrane Database Syst Rev. 2015 Oct 22;(10):CD005328. doi: 10.1002/14651858.CD005328.pub3. PMID: 16625636 Updated.
-
Viscosupplementation for the treatment of osteoarthritis of the knee.Cochrane Database Syst Rev. 2006 Apr 19;2006(2):CD005321. doi: 10.1002/14651858.CD005321.pub2. Cochrane Database Syst Rev. 2006. PMID: 16625635 Free PMC article.
References
-
- Kucharz EJ, Szántó S, Ivanova Goycheva M, et al. Endorsement by Central European experts of the revised ESCEO algorithm for the management of knee osteoarthritis. Rheumatol Int. 2019;39:1117–23. - PubMed
-
- Balazs EA, Denlinger JL. Viscosupplementation: a new concept in the treatment of osteoarthritis. J Rheumatol Suppl. 1993;39:3–9. - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

