A paternal lactate dehydrogenase critically enhances male gametogenesis and malaria transmission
- PMID: 40603939
- PMCID: PMC12223287
- DOI: 10.1038/s41598-025-05832-1
A paternal lactate dehydrogenase critically enhances male gametogenesis and malaria transmission
Abstract
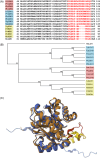
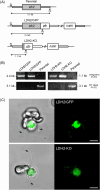
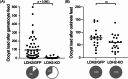
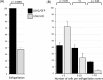
Malaria blood stage parasite development relies on glycolysis to generate ATP, which requires pyruvate to lactate conversion by an essential lactate dehydrogenase enzyme (LDH1). Conversely, parasites developing in the mosquito employ mitochondrial chemiosmosis for ATP production. The source of ATP during transition from vertebrate to insect is less clear; gametes form in the mosquito midgut lumen within minutes of gametocyte ingestion, and while female gametes possess a mitochondrion, this organelle is absent from male gametes (microgametes). Here, we investigate a second LDH enzyme (LDH2) found exclusively in male gametocytes and microgametes. Knockout of Plasmodium berghei LDH2 expression reduces the number and size of exflagellation centres and radically diminishes oocyst development in Anopheles stephensi mosquitoes. Our data indicate that LDH2 supplements LDH1 activity to facilitate the cytokinesis step of male gametogenesis, while LDH1 alone is sufficient for motility of free-swimming microgametes. Our results point to a key role for glycolytic ATP production in microgamete formation and function and identify LDH activity as a potential malaria transmission-blocking drug target.
Keywords: Gametogenesis; Glycolysis; Lactate dehydrogenase; Plasmodium; Transmission-blocking drugs.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures
Similar articles
-
Plasmodium falciparum PP1 phosphatase is a key regulator of malaria parasite transmission.mBio. 2025 Aug 18:e0087425. doi: 10.1128/mbio.00874-25. Online ahead of print. mBio. 2025. PMID: 40823845
-
Primaquine for reducing Plasmodium falciparum transmission.Cochrane Database Syst Rev. 2012 Sep 12;(9):CD008152. doi: 10.1002/14651858.CD008152.pub2. Cochrane Database Syst Rev. 2012. Update in: Cochrane Database Syst Rev. 2014 Jun 30;(6):CD008152. doi: 10.1002/14651858.CD008152.pub3. PMID: 22972117 Updated.
-
Larvivorous fish for preventing malaria transmission.Cochrane Database Syst Rev. 2017 Dec 11;12(12):CD008090. doi: 10.1002/14651858.CD008090.pub3. Cochrane Database Syst Rev. 2017. PMID: 29226959 Free PMC article.
-
An atypical Arp2/3 complex is required for Plasmodium DNA segregation and malaria transmission.Nat Microbiol. 2025 Jul;10(7):1775-1790. doi: 10.1038/s41564-025-02023-6. Epub 2025 Jun 13. Nat Microbiol. 2025. PMID: 40514561 Free PMC article.
-
Loss of serine/threonine protein phosphatase 6 severely impairs sexual stage development in malaria parasite Plasmodium berghei.PLoS Pathog. 2025 Jul 7;21(7):e1013318. doi: 10.1371/journal.ppat.1013318. eCollection 2025 Jul. PLoS Pathog. 2025. PMID: 40623036 Free PMC article.
References
-
- Painter, H. J., Morrisey, J. M., Mather, M. W. & Vaidya, A. B. Specific role of mitochondrial electron transport in blood-stage Plasmodium falciparum. Nature446, 88–91. 10.1038/nature05572 (2007). - PubMed
-
- Royer, R. E., Deck, L. M., Campos, N. M., Hunsaker, L. A. & Vander jagt, D. L. Biologically active derivatives of gossypol: Synthesis and antimalarial activities of peri-acylated gossylic nitriles. J. Med. Chem.29, 1799–1801. 10.1021/jm00159a043 (1986). - PubMed
-
- Dunn, C. R. et al. The structure of lactate dehydrogenase from Plasmodium falciparum reveals a new target for anti-malarial design. Nat. Struct. Biol.3, 912–915. 10.1038/nsb1196-912 (1996). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

