FAM72A promotes UNG2 degradation and mutagenesis in human cancer cells
- PMID: 40604025
- PMCID: PMC12223117
- DOI: 10.1038/s41598-025-07723-x
FAM72A promotes UNG2 degradation and mutagenesis in human cancer cells
Abstract
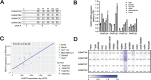
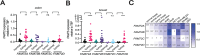
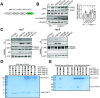
Genetic lesions drive cancer development and progression, and understanding their origins will reveal the mechanisms of carcinogenesis. We showed that murine FAM72A promotes mutagenic DNA repair during antibody maturation by acting as a substrate adaptor of the CTLHMKLN1 E3 ligase to induce the proteasome degradation of Uracil DNA glycosylase 2 (UNG2), a pivotal enzyme of the base excision repair. In humans, the FAM72 gene has expanded to include four paralogues named FAM72A-D. Bioinformatic studies suggested that the human FAM72 genes are overexpressed in a broad range of cancers. However, the functional roles of FAM72A-D in human biology and cancer are unknown. Here, we show that FAM72 family members are minimally expressed in most healthy tissues except for thymus, and that FAM72A, B and D are overexpressed in primary tumorigenic tissues. Human FAM72 expression inversely correlates with UNG2 protein level in human cell lines and primary tumorigenic tissues suggesting that human FAM72 promotes UNG2 degradation. However, only FAM72A is able to bind to and induce UNG2 degradation in human cells. Our results suggest that the ability of FAM72A to induce UNG2 degradation contributes to neoplasia in a variety of cancer types by promoting mutagenic repair of genomic dUs.
Keywords: Base excision repair; Cancer; Cytosine deamination; FAM72; Mutation; Uracil DNA glycosylase 2.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Conflict of interest: F.S is a founder and consultant for Repare Therapeutics and Induxion Therapeutics.
Figures

References
-
- Methot, S. P. & Di Noia, J. M. Molecular mechanisms of somatic hypermutation and class switch recombination. Adv. Immunol.133, 37–87. 10.1016/bs.ai.2016.11.002 (2017). - PubMed
-
- Cascalho, M., Wong, J., Steinberg, C. & Wabl, M. Mismatch repair co-opted by hypermutation. Science279, 1207–1210 (1998). - PubMed
-
- Martin, A. & Scharff, M. D. AID and mismatch repair in antibody diversification. Nat. Rev. Immunol.2, 605–614 (2002). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

