Immune correlates of anti-BCMA CAR-T products idecabtagene vicleucel and ciltacabtagene autoleucel in a real-world cohort of patients with multiple myeloma
- PMID: 40610432
- PMCID: PMC12229333
- DOI: 10.1038/s41467-025-60980-2
Immune correlates of anti-BCMA CAR-T products idecabtagene vicleucel and ciltacabtagene autoleucel in a real-world cohort of patients with multiple myeloma
Abstract
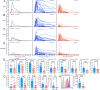
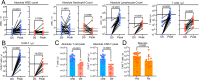
We performed the first in-depth, comparative and prospective biomonitoring of Multiple Myeloma (MM) patients (N = 39) receiving ciltacabtagene autoleucel (cilta-cel) or idecabtagene vicleucel (ide-cel) chimeric antigen receptor T cells (CAR T) in the real-world setting. In cilta-cel patients response rates were higher and atypical neurotoxicities/infections more frequent. Peak CAR T counts were significantly higher in cilta-cel patients, driven by CD4+ CAR expansion, correlating with clinical responses. Expansion of cilta-cel cells was associated with higher CAR and CD27 expression while, in contrast to ide-cel, there was no correlation between TIM3 expression and CAR T proliferation. Cilta-cel CAR T expansion was followed by a CAR-specific switch from proliferation-associated genes to genes/surface markers indicating effector/memory function. The longer persistence of cilta-cel CAR T was associated with increased IL-7R expression; in vitro data showed persistent antigen-independent activation and higher metabolic activity of cilta-cel vs. ide-cel CAR T. Among cilta-cel-treated patients experiencing atypical neurotoxicities, central nervous system (CNS)-infiltrating, effector-type CAR T presented a distinct inflammatory phenotypic/cytokine-expression profile. This in-depth biomonitoring report following real-world cilta-cel or ide-cel highlights intrinsic biological differences between BCMA-targeting CAR T products, potentially explaining differences in clinical activity and toxicity. Our findings may guide optimization of cellular immunotherapy strategies in MM.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: T.L. receives a salary from AbbVie. The other authors declare that they do not have any competing interests.
Figures




References
-
- Munshi, N. C. et al. Idecabtagene Vicleucel in Relapsed and Refractory Multiple Myeloma. N. Engl. J. Med.384, 705–716 (2021). - PubMed
-
- Berdeja, J. G. et al. Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): a phase 1b/2 open-label study. Lancet398, 314–324 (2021). - PubMed
-
- Inxight Drugs - CILTACABTAGENE AUTOLEUCEL TRANSGENE INSERT. https://drugs.ncats.io/substance/7S3Z5YP744 (2024).
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

