Overexpression of the STOX1B isoform of STOX1 triggers preeclampsia-like symptoms through HNF4α-dependent alterations of coagulation cascades in mice
- PMID: 40610507
- PMCID: PMC12229339
- DOI: 10.1038/s41598-025-07731-x
Overexpression of the STOX1B isoform of STOX1 triggers preeclampsia-like symptoms through HNF4α-dependent alterations of coagulation cascades in mice
Abstract
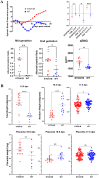
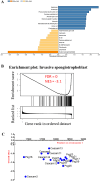
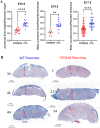
STOX1 has been involved in genetic forms of preeclampsia. The gene encodes two major isoforms coined STOX1A and STOX1B (989 and 227 amino-acids, respectively), sharing the same DNA binding domain. The two isoforms have opposite function on major genes involved in trophoblast syncytialization and oxidative stress management. Placenta-fetal overexpression of STOX1A induces preeclampsia in mice. Here we explore the effects of STOX1B placenta-fetal overexpression. A STOX1B transgenic mouse line (expression restricted to the foeto-placental unit through ad hoc crosses) was analyzed in terms of blood pressure, proteinuria, soluble antiangiogenic factors, placental and fetal weights, maternal heart weight, and placental histology. Placental gene expression was explored by RNA-sequencing, followed by a bioinformatics analysis. Female mice carrying STOX1B placentas displayed preeclampsia features with a striking increase of genes involved in coagulation, presumably under the control of the HNF4α transcription factor. Genes specific of the spongiotrophoblast were strongly down-regulated consistently with a junctional zone reduction. This new model of preeclampsia seems connected with an enhancement of coagulation processes, similar to preeclamptic patients living at high altitude. Our model could be useful to study the features of preeclampsia in this context, and be a convenient complement to other animal models of preeclampsia.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures


References
-
- Erlandsson, L. et al. Inventory of novel animal models addressing etiology of preeclampsia in the development of new therapeutic/intervention opportunities. Am. J. Reprod. Immunol.75, 402–410 (2016). - PubMed
-
- McCarthy, F. P., Kingdom, J. C., Kenny, L. C. & Walsh, S. K. Animal models of preeclampsia; uses and limitations. Placenta32, 413–419 (2011). - PubMed
-
- van Dijk, M. et al. Maternal segregation of the Dutch preeclampsia locus at 10q22 with a new member of the winged helix gene family. Nat. Genet.37, 514–519 (2005). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

