Disruption of normal stem cell function and transmission of myelodysplastic syndrome by self-renewal of committed myeloid lineage cells
- PMID: 40614733
- PMCID: PMC12365822
- DOI: 10.1016/j.stemcr.2025.102571
Disruption of normal stem cell function and transmission of myelodysplastic syndrome by self-renewal of committed myeloid lineage cells
Abstract
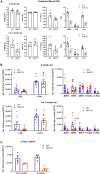
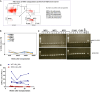
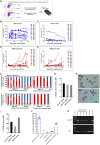
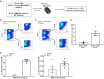
The ineffective hematopoiesis of myelodysplastic syndrome (MDS) suggests that hematopoietic stem and progenitor cells (HSPCs) are defective. Here, we demonstrate that NUP98::HOXD13 (NHD13) MDS mice have significantly decreased functional HSPCs. Moreover, in contrast to wild-type (WT) bone marrow (BM), lineage-positive (Lin+) BM cells from NHD13 mice have self-renewal potential. Specific subsets of NHD13 Lin+ cells that express B220 and Kit antigens were able to self-renew and generate MDS in WT recipients. Although this unique B220+Kit+ phenotype could be found in WT as well as NHD13 BM, the population was markedly increased in NHD13 BM. Further characterization using Mac1 and Gr1 markers revealed that both Mac1+Gr1+B220+Kit+ and Mac1-Gr1- B220+Kit+ populations showed self-renewal and led to an MDS phenotype in WT recipients. Taken together, these findings demonstrate that as normal hematopoiesis derived from typical HSPCs decreases in NHD13 mice, committed hematopoietic progenitor cells proliferate, self-renew, and initiate MDS.
Keywords: MDS-initiating cell; NHD13; hematopoiesis; kit; myelodysplastic syndrome; stem cells.
Published by Elsevier Inc.
Conflict of interest statement
Declaration of interests P.D.A. receives royalties from the NIH Technology Transfer program for the invention of NHD13 mice.
Figures


References
-
- Balderman S.R., Li A.J., Hoffman C.M., Frisch B.J., Goodman A.N., LaMere M.W., Georger M.A., Evans A.G., Liesveld J.L., Becker M.W., Calvi L.M. Targeting of the bone marrow microenvironment improves outcome in a murine model of myelodysplastic syndrome. Blood. 2016;127:616–625. doi: 10.1182/blood-2015-06-653113. - DOI - PMC - PubMed
-
- Benito A.I., Bryant E., Loken M.R., Sale G.E., Nash R.A., John Gass M., Deeg H.J. NOD/SCID mice transplanted with marrow from patients with myelodysplastic syndrome (MDS) show long-term propagation of normal but not clonal human precursors. Leuk. Res. 2003;27:425–436. doi: 10.1016/S0145-2126(02)00221-7. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

