Airway epithelial stem cell renewal and differentiation: overcoming challenging steps towards clinical-grade tissue engineering
- PMID: 40619403
- PMCID: PMC12232793
- DOI: 10.1186/s13287-025-04478-0
Airway epithelial stem cell renewal and differentiation: overcoming challenging steps towards clinical-grade tissue engineering
Abstract
Background: Despite their life-saving potential, tissue engineering approaches for the treatment of extensive tracheal and bronchial defects still face significant limitations. A major challenge is the inability to regenerate a functional airway epithelium containing the appropriate amount of stem cells required for long-term tissue renewal following transplantation of the bioengineered graft. In this scenario, extensive cell culture characterization, validation assays and quality controls are needed to guide each step of the regeneration process.
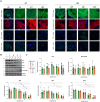
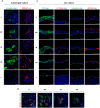
Methods: Stem cell depletion is often due to suboptimal culture conditions, therefore we tested the ability of a clinical-grade culture system to support the safe and efficient in vitro expansion and differentiation of primary human tracheal and bronchial epithelial cells. Single-cell clonal analysis was used to unravel the heterogeneity of airway basal cells and to understand tissue-specific regeneration and differentiation mechanisms. Functional assays were used to investigate the wound healing ability and tightness of the regenerated epithelium under the selected culture conditions.
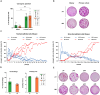
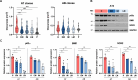
Results: Primary tracheobronchial epithelial cells showed an impressive proliferative potential, allowing the regeneration of a mature and functional epithelium without immortalisation events. Analysis at the single cell level allowed the identification of the subpopulation of basal cells endowed with in vitro self-renewal, distinguishing them from transient amplifying cells. This approach has further defined the hierarchy of cellular differentiation and its correlation with regenerative and differentiation potential.
Conclusions: Our results show that primary airway epithelial cell cultures can maintain stem cells together with their differentiation lineages in vitro. Airway cells can be safely and effectively used in autologous tissue engineering approaches when cultured under appropriate and well-standardised conditions. In addition to the validation assays proposed for the development of new advanced therapy products, this study outlines possible quality controls to enhance therapeutic success and maximise patient safety in future clinical applications.
Keywords: Airway differentiation; Airway holoclones; Airway reconstruction; Clinical-grade expansion; Epithelial stem cells; Regenerative medicine; Respiratory mucosa; Stem cell markers; Tissue engineering; Tracheobronchial epithelium.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethical approval and consent to participate: All specimens were obtained in accordance with the tenets of the Declaration of Helsinki and anonymized. Human tracheal and bronchial samples were collected from n = 5 males and n = 4 female donors aged between 38 and 76 years. The donors provided informed consent for the use of their biological material for the present study, and ethical committee approval was obtained from all involved centres “Arcispedale S. Maria Nuova di Reggio Emilia”, IRCCS, Reggio Emilia, Italy, (title “Pre-clinical study aimed at characterizing human respiratory epithelial stem cells, their differentiation pathways and potential use as an in vitro model for toxicity studies and tissue engineering”; “Comitato Etico Area Vasta Emilia Nord”, Protocol N. 2019/0014725; date of approval 05/02/2019), and the “Fondazione Policlinico Universitario A. Gemelli”, IRCCS, Rome, Italy (title “Pre-clinical study aimed at characterizing human respiratory epithelial stem cells, their differentiation pathways and potential use as an in vitro model for toxicity studies and tissue engineering”; “Comitato Etico Territoriale Lazio Area 3” N. 0008968/21; date of approval 10/03/2021). Human skin samples were obtained from “Azienda Ospedaliero-Universitaria di Modena”, IRCSS, Modena, Italy” from healthy living donors upon informed consent and compliance with Italian regulations (title “Human epithelial stem cells: characterization and development of clinical applications in Regenerative Medicine”; Comitato Etico dell’Area Vasta Emilia Nord, number 178/09; date of approval 29/09/2009). Consent for publication: All the authors provided consent for publication. Competing interests: The authors declare no competing interests.
Figures




References
-
- Maina JN. Functional morphology of the avian respiratory system, the lung-air sac system: efficiency built on complexity. Ostrich. 2008;79(2):117–32.
-
- Bhutani VK. Pulmonary gas exchange. Manual of neonatal respiratory care. Boston, MA: Springer US; 2012. pp. 39–47.
-
- Lawrence DA, Branson B, Oliva I, Rubinowitz A. The wonderful world of the windpipe: a review of central airway anatomy and pathology. Can Assoc Radiol J. 2015;66(1):30–43. - PubMed
-
- Piazza C, Filauro M, Dikkers FG, Nouraei SAR, Sandu K, Sittel C, et al. Long-term intubation and high rate of tracheostomy in COVID-19 patients might determine an unprecedented increase of airway stenoses: a call to action from the European laryngological society. Eur Arch Otorhinolaryngol. 2021;278(1):1–7. - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

