Comparison of homologous and heterologous vaccination strategies for combating disease caused by Burkholderia pseudomallei
- PMID: 40621462
- PMCID: PMC12226310
- DOI: 10.3389/fimmu.2025.1596265
Comparison of homologous and heterologous vaccination strategies for combating disease caused by Burkholderia pseudomallei
Abstract
Introduction: Melioidosis is a major cause of disease and mortality in endemic tropical regions, and the etiologic agent, Burkholderia pseudomallei, is being isolated increasingly from an expanded range of environmental and clinical sources in locations including the United States. The disease can have multi-faceted clinical presentations and requires a complex and protracted treatment regimen which is confounded by resistance of this microbe to numerous antibiotics. Thus, prophylactic countermeasures are needed; however, a vaccine has yet to be licensed for human use. Since B. pseudomallei is classified as a Tier 1 select agent, the development of a safe and effective vaccine is both a military and public health need. Our laboratories have focused on the development of vaccines composed of live attenuated strains and defined subunit antigens.
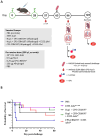
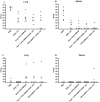
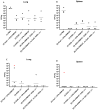
Methods: In the current study, we evaluated homologous and heterologous combinations of candidate subunits and live vaccines in a murine aerosol model of melioidosis to determine the effects of vaccine composition and delivery scheme on protection in conjunction with immune responses and bacterial clearance.
Results: Both strategies provided significant protection against lethal aerosol challenges, and the accumulated data support that a heterologous vaccination strategy employing capsular polysaccharide conjugate and Hcp1 subunits and a live but highly safe capsular polysaccharide-producing surrogate strain of B. thailandensis is an effective and potentially agile prophylactic strategy.
Keywords: Burkholderia pseudomallei; heterologous; immunity; melioidosis; mice; protection; vaccines.
Copyright © 2025 Biryukov, Cote, Klimko, Dankmeyer, Rill, Hunter, Davies, Hedrick, Shoe, Schmidt, Orne, Qiu, Welkos, Burtnick, Brett and DeShazer.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

