Rh-Catalyzed Atroposelective Single-Carbon Insertion
- PMID: 40621830
- PMCID: PMC12272680
- DOI: 10.1021/jacs.5c06139
Rh-Catalyzed Atroposelective Single-Carbon Insertion
Abstract
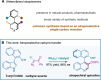
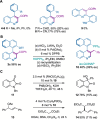
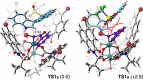
Single-carbon insertion processes have gained considerable momentum over the past few years. Although innovative methods have emerged for converting indole or indene into quinoline or naphthalene cores, the enantioselective version of such ring-expansions to create (hetero)biaryl atropisomers has not been developed. Herein, we report the first enantioselective single-carbon insertion that converts 3-aryl indoles to atropochiral quinolines. Key in the process was the generation of a chiral Rh-carbynoid that mediated in the creation of the stereogenic C(sp2)-C(sp2) axis.
Figures

References
-
-
For selected reviews:
- Bringmann G., Price Mortimer A. J., Keller P. A., Gresser M. J., Garner J., Breuning M.. Atroposelective Synthesis of Axially Chiral Biaryl Compounds. Angew. Chem., Int. Ed. 2005;44:5384–5427. doi: 10.1002/anie.200462661. - DOI - PubMed
- Kozlowski M. C., Morgan B. J., Linton E. C.. Total Synthesis of Chiral Biaryl Natural Products by Asymmetric Biaryl Coupling. Chem. Soc. Rev. 2009;38:3193–3207. doi: 10.1039/b821092f. - DOI - PMC - PubMed
- Bringmann G., Gulder T., Gulder T. A. M., Breuning M.. Atroposelective Total Synthesis of Axially Chiral Biaryl Natural Products. Chem. Rev. 2011;111:563–639. doi: 10.1021/cr100155e. - DOI - PubMed
- LaPlante S. R., Fader L. D., Fandrick K. R., Fandrick D. R., Hucke O., Kemper R., Miller S. P. F., Edwards P. J.. Assessing Atropisomer Axial Chirality in Drug Discovery and Development. J. Med. Chem. 2011;54:7005–7022. doi: 10.1021/jm200584g. - DOI - PubMed
- Smyth J. E., Butler N. M., Keller P. A.. A Twist of Nature–the Significance of Atropisomers in Biological Systems. Nat. Prod. Rep. 2015;32:1562–1583. doi: 10.1039/C4NP00121D. - DOI - PubMed
-
See also:
- O’Connell P. J., Harms C. T., Allen J. R. F.. Metolachlor, S-Metolachlor and Their Role within Sustainable Weed-Management. Crop Prot. 1998;17:207–212. doi: 10.1016/S0261-2194(98)80011-2. - DOI
- Degenhardt C. F., Lavin J. M., Smith M. D., Shimizu K. D.. Conformationally Imprinted Receptors: Atropisomers with “Write”, “Save”, and “Erase” Recognition Properties. Org. Lett. 2005;7:4079–4081. doi: 10.1021/ol051325t. - DOI - PubMed
- Zhang D.-W., Li M., Chen C.-F.. Recent Advances in Circularly Polarized Electroluminescence Based on Organic Light-Emitting Diodes. Chem. Soc. Rev. 2020;49:1331–1343. doi: 10.1039/C9CS00680J. - DOI - PubMed
- Hamada H., Itabashi Y., Shang R., Nakamura E.. Axially Chiral Spiro-Conjugated Carbon-Bridged p-Phenylenevinylene Congeners: Synthetic Design and Materials Properties. J. Am. Chem. Soc. 2020;142:2059–2067. doi: 10.1021/jacs.9b13019. - DOI - PubMed
-
-
- Cheng J. K., Xiang S.-H., Li S., Ye L., Tan B.. Recent Advances in Catalytic Asymmetric Construction of Atropisomers. Chem. Rev. 2021;121:4805–4902. doi: 10.1021/acs.chemrev.0c01306. - DOI - PubMed
- Liu C.-X., Zhang W.-W., Yin S.-Y., Gu Q., You S.-L.. Synthesis of Atropisomers by Transition-Metal-Catalyzed Asymmetric C–H Functionalization Reactions. J. Am. Chem. Soc. 2021;143:14025–14040. doi: 10.1021/jacs.1c07635. - DOI - PubMed
- Rodríguez-Salamanca P., Fernández R., Hornillos V., Lassaletta J. M.. Asymmetric Synthesis of Axially Chiral C–N Atropisomers. Chem.Eur. J. 2022;28:e202104442. doi: 10.1002/chem.202104442. - DOI - PMC - PubMed
- Roos C. B., Chiang C.-H., Murray L. A. M., Yang D., Schulert L., Narayan A. R. H.. Stereodynamic Strategies to Induce and Enrich Chirality of Atropisomers at a Late Stage. Chem. Rev. 2023;123:10641–10727. doi: 10.1021/acs.chemrev.3c00327. - DOI - PubMed
- Schmidt T. A., Hutskalova V., Sparr C.. Atroposelective Catalysis. Nat. Rev. Chem. 2024;8:497–517. doi: 10.1038/s41570-024-00618-x. - DOI - PubMed
- Zhang H., Li T., Liu S., Shi F.. Catalytic Asymmetric Synthesis of Atropisomers Bearing Multiple Chiral Elements: An Emerging Field. Angew. Chem., Int. Ed. 2024;63:e202311053. doi: 10.1002/anie.202311053. - DOI - PubMed
- Xiang S.-H., Ding W.-Y., Wang Y.-B., Tan B.. Catalytic Atroposelective Synthesis. Nat. Catal. 2024;7:483–498. doi: 10.1038/s41929-024-01138-z. - DOI
-
- Liu Y., Tse Y.-L. S., Kwong F. Y., Yeung Y.-Y.. Accessing Axially Chiral Biaryls via Organocatalytic Enantioselective Dynamic-Kinetic Resolution-Semipinacol Rearrangement. ACS Catal. 2017;7:4435–4440. doi: 10.1021/acscatal.7b01056. - DOI
-
- Tang X., Tang Y., Peng J., Du H., Huang L., Gao J., Liu S., Wang D., Wang W., Gao L., Lan Y., Song Z.. Ligand-Controlled Regiodivergent Ring Expansion of Benzosilacyclobutenes with Alkynes En Route to Axially Chiral Silacyclohexenyl Arenes. J. Am. Chem. Soc. 2024;146:26639–26648. doi: 10.1021/jacs.4c00252. - DOI - PubMed
-
- Ciamician G. L., Dennstedt M.. Ueber die Einwirkung des Chloroforms auf die Kaliumverbindung Pyrrols. Berichte Dtsch. Chem. Ges. 1881;14:1153–1163. doi: 10.1002/cber.188101401240. - DOI
- Magnanini G.. Ueber die Acetylverbindungen des Methylketols und des Skatols. Berichte Dtsch. Chem. Ges. 1888;21:1936–1939. doi: 10.1002/cber.188802101373. - DOI
- Rees C. W., Smithen C. E.. 181. The Mechanism of Heterocyclic Ring Expansions. Part I. The Reaction of 2,3-Dimethylindole with Dichlorocarbene. J. Chem. Soc. Resumed. 1964:928–937. doi: 10.1039/jr9640000928. - DOI
- Dobbs H. E.. Radiotracer Study of the Reaction of Dichloro-Carbene with Dimethylindole. Tetrahedron. 1968;24:491–496. doi: 10.1016/0040-4020(68)89048-9. - DOI
- Jones R. L., Rees C. W.. Mechanism of Heterocyclic Ring Expansions. Part III. Reaction of Pyrroles with Dichlorocarbene. J. Chem. Soc. C Org. 1969;18:2249–2251. doi: 10.1039/j39690002249. - DOI
- Ogawa H., Aoyama T., Shioiri T.. Lithium Trimethylsilyldiazomethane: A Convenient Reagent for the Preparation of Cyclohepta[b]Pyrrol-2-Ones from N-Methylanilides of α-Keto Acids. Synlett. 1994;1994:757–758. doi: 10.1055/s-1994-22999. - DOI
- Morita M., Hari Y., Iguchi T., Aoyama T.. Facile Synthesis of 2-Azaazulenes from Thiobenzoyl Isocyanates Using Trimethylsilyldiazomethane. Tetrahedron. 2008;64:1753–1758. doi: 10.1016/j.tet.2007.11.107. - DOI
- Morita M., Hari Y., Aoyama T.. Facile Synthesis of 1-Methyl-1H-Benzo[b]Azepines from 1-Methylquinolinium Iodides and Diazo(Trimethylsilyl)Methylmagnesium Bromide. Synthesis. 2010;2010:4221–4227. doi: 10.1055/s-0030-1258306. - DOI
- Mortén M., Hennum M., Bonge-Hansen T.. Synthesis of Quinoline-3-Carboxylates by a Rh(II)-Catalyzed Cyclopropanation-Ring Expansion Reaction of Indoles with Halodiazoacetates. Beilstein J. Org. Chem. 2015;11:1944–1949. doi: 10.3762/bjoc.11.210. - DOI - PMC - PubMed
- Wang H., Zhou C., Che C.. Cobalt-Porphyrin-Catalyzed Intramolecular Buchner Reaction and Arene Cyclopropanation of In Situ Generated Alkyl Diazomethanes. Adv. Synth. Catal. 2017;359:2253–2258. doi: 10.1002/adsc.201700205. - DOI
- Peeters S., Berntsen L. N., Rongved P., Bonge-Hansen T.. Cyclopropanation–Ring Expansion of 3-Chloroindoles with α-Halodiazoacetates: Novel Synthesis of 4-Quinolone-3-Carboxylic Acid and Norfloxacin. Beilstein J. Org. Chem. 2019;15:2156–2160. doi: 10.3762/bjoc.15.212. - DOI - PMC - PubMed
- Wang Z., Jiang L., Sarró P., Suero M. G.. Catalytic Cleavage of C(sp 2)–C(sp 2) Bonds with Rh-Carbynoids. J. Am. Chem. Soc. 2019;141:15509–15514. doi: 10.1021/jacs.9b08632. - DOI - PubMed
- Dherange B. D., Kelly P. Q., Liles J. P., Sigman M. S., Levin M. D.. Carbon Atom Insertion into Pyrroles and Indoles Promoted by Chlorodiazirines. J. Am. Chem. Soc. 2021;143:11337–11344. doi: 10.1021/jacs.1c06287. - DOI - PMC - PubMed
- Kim J., Yoo E. J.. Catalytic Ring Expansion of Activated Heteroarenes Enabled by Regioselective Dearomatization. Org. Lett. 2021;23:4256–4260. doi: 10.1021/acs.orglett.1c01173. - DOI - PubMed
- Ma D., Martin B. S., Gallagher K. S., Saito T., Dai M.. One-Carbon Insertion and Polarity Inversion Enabled a Pyrrole Strategy to the Total Syntheses of Pyridine-Containing Lycopodium Alkaloids: Complanadine A and Lycodine. J. Am. Chem. Soc. 2021;143:16383–16387. doi: 10.1021/jacs.1c08626. - DOI - PMC - PubMed
- Jiang L., Sarró P., Teo W. J., Llop J., Suero M. G.. Catalytic Alkene Skeletal Modification for the Construction of Fluorinated Tertiary Stereocenters. Chem. Sci. 2022;13:4327–4333. doi: 10.1039/D2SC00968D. - DOI - PMC - PubMed
- Piacentini P., Bingham T. W., Sarlah D.. Dearomative Ring Expansion of Polycyclic Arenes. Angew. Chem., Int. Ed. 2022;61:e202208014. doi: 10.1002/anie.202208014. - DOI - PMC - PubMed
- Joynson B. W., Cumming G. R., Ball L. T.. Photochemically Mediated Ring Expansion of Indoles and Pyrroles with Chlorodiazirines: Synthetic Methodology and Thermal Hazard Assessment. Angew. Chem., Int. Ed. 2023;62:e202305081. doi: 10.1002/anie.202305081. - DOI - PMC - PubMed
- Park S., Kim C.-E., Jeong J., Ryu H., Maeng C., Kim D., Baik M.-H., Lee P. H.. Selective Ring Expansion and C–H Functionalization of Azulenes. Nat. Commun. 2023;14:7936. doi: 10.1038/s41467-023-43200-7. - DOI - PMC - PubMed
- Guo H., Qiu S., Xu P.. One-Carbon Ring Expansion of Indoles and Pyrroles: A Straightforward Access to 3-Fluorinated Quinolines and Pyridines. Angew. Chem., Int. Ed. 2024;63:e202317104. doi: 10.1002/anie.202317104. - DOI - PubMed
- Lamb J. S., Koyama F., Suzuki N., Suzuki Y.. Carbon Atom Insertion into N -Heterocyclic Carbenes to Yield 3,4-Dihydroquinoxalin-2(1H)-Ones. Org. Chem. Front. 2024;11:277–283. doi: 10.1039/D3QO01579C. - DOI
- Li C., Chen L., Wang H., Yan Z., Lyu B., Lyu W., Jiang C., Lu D., Li J., Jiao N., Song S.. CF Bond Insertion into Indoles with CHBr2F: An Efficient Method to Synthesize Fluorinated Quinolines and Quinolones. Chin. J. Chem. 2024;42:1128–1132. doi: 10.1002/cjoc.202400033. - DOI
- Timmann S., Wu T.-H., Golz C., Alcarazo M.. Reactivity of α-Diazo Sulfonium Salts: Rhodium-Catalysed Ring Expansion of Indenes to Naphthalenes. Chem. Sci. 2024;15:5938–5943. doi: 10.1039/D4SC01138D. - DOI - PMC - PubMed
- Wu F.-P., Chintawar C. C., Lalisse R., Mukherjee P., Dutta S., Tyler J., Daniliuc C. G., Gutierrez O., Glorius F.. Ring Expansion of Indene by Photoredox-Enabled Functionalized Carbon-Atom Insertion. Nat. Catal. 2024;7:242–251. doi: 10.1038/s41929-023-01089-x. - DOI - PMC - PubMed
- Wu F.-P., Tyler J. L., Daniliuc C. G., Glorius F.. Atomic Carbon Equivalent: Design and Application to Diversity-Generating Skeletal Editing from Indoles to 3-Functionalized Quinolines. ACS Catal. 2024;14:13343–13351. doi: 10.1021/acscatal.4c03868. - DOI
- Li L., Chen H., Zhang X., Murali K., Zhu Q., Liu M., Zhang H., Nenajdenko V., Bi X.. Silver-Catalyzed Single-Carbon Insertion of Indoles with Acetophenone N-Triftosylhydrazones. Org. Lett. 2024;26:7207–7211. doi: 10.1021/acs.orglett.4c02633. - DOI - PubMed
- Liu S., Yang Y., Song Q., Liu Z., Sivaguru P., Zhang Y., De Ruiter G., Anderson E. A., Bi X.. Halogencarbene-Free Ciamician-Dennstedt Single-Atom Skeletal Editing. Nat. Commun. 2024;15:9998. doi: 10.1038/s41467-024-54379-8. - DOI - PMC - PubMed
- Liu S., Yang Y., Song Q., Liu Z., Lu Y., Wang Z., Sivaguru P., Bi X.. Tunable Molecular Editing of Indoles with Fluoroalkyl Carbenes. Nat. Chem. 2024;16:988–997. doi: 10.1038/s41557-024-01468-2. - DOI - PubMed
- Liu L., Tian M., Lang Z., Wang Y., He C., Chen Y., Han W.. Indole-Quinoline Transmutation Enabled by a Formal Rhodium Carbynoid. Angew. Chem. Int. Ed. 2025;64:e202501966. doi: 10.1002/anie.202501966. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

