Recent advances of engineering cell membranes for nanomedicine delivery across the blood-brain barrier
- PMID: 40624508
- PMCID: PMC12235962
- DOI: 10.1186/s12951-025-03572-y
Recent advances of engineering cell membranes for nanomedicine delivery across the blood-brain barrier
Abstract
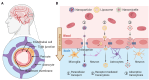
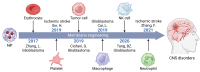
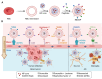
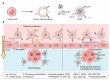
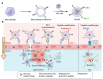
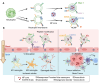
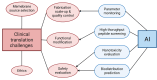
The blood-brain barrier (BBB) poses a major challenge to the effective delivery of therapeutic agents for the treatment of central nervous system (CNS) disorders. The integration of cell membrane engineering with nanotechnology has recently enabled the development of membrane-engineered nanoparticles (CNPs). These nanocarriers exhibit enhanced BBB penetration and improved CNS targeting. This review systematically summarizes the latest advances in the development and application of CNPs, emphasizing how different cellular sources-such as erythrocytes, platelets, tumor cells, and leukocytes-impact delivery efficiency and therapeutic outcomes. We also examine the molecular mechanisms underlying nanoparticle-BBB interactions and highlight the importance of biosafety evaluations. Moreover, critical barriers to clinical translation, including large-scale manufacturing challenges, batch-to-batch variability, and regulatory complexities, are discussed. Finally, we explore emerging strategies-particularly the integration of artificial intelligence (AI)-that hold potential for overcoming existing clinical gaps, enabling the rational design and optimized development of CNP-based therapeutics for CNS disorders. By integrating mechanistic insights with translational perspectives, this review provides a clear conceptual and technological foundation for the development of next-generation CNS-targeted nanotherapeutics.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: All the authors have read and approved the manuscript. Competing interests: The authors declare no competing interests.
Figures


Similar articles
-
Overcoming the blood-brain barrier (BBB) in pediatric CNS tumors: immunotherapy and nanomedicine-driven strategies.Med Oncol. 2025 Aug 19;42(10):431. doi: 10.1007/s12032-025-02984-y. Med Oncol. 2025. PMID: 40828479 Review.
-
Cell membrane-coated nanoparticles for neurodegenerative disorders management.Int J Pharm. 2025 Aug 20;681:125875. doi: 10.1016/j.ijpharm.2025.125875. Epub 2025 Jun 20. Int J Pharm. 2025. PMID: 40544973 Review.
-
Ultrasmall nanoparticles for co-delivery of antisense oligonucleotides targeting miR-21 and miR-210 to treat glioblastoma.J Nanobiotechnology. 2025 Jul 2;23(1):482. doi: 10.1186/s12951-025-03529-1. J Nanobiotechnology. 2025. PMID: 40604820 Free PMC article.
-
Framework Nucleic Acid Nanomaterials for Central Nervous System Therapies: Design for Barrier Penetration, Targeted Delivery, Cellular Uptake, and Endosomal Escape.ACS Nano. 2025 Jul 15;19(27):24335-24376. doi: 10.1021/acsnano.5c03945. Epub 2025 Jun 30. ACS Nano. 2025. PMID: 40587770 Review.
-
Exosomes as nanocarriers for brain-targeted delivery of therapeutic nucleic acids: advances and challenges.J Nanobiotechnology. 2025 Jun 18;23(1):453. doi: 10.1186/s12951-025-03528-2. J Nanobiotechnology. 2025. PMID: 40533746 Free PMC article. Review.
References
-
- Park SH, Kim MJ, Jung HH, Chang WS, Choi HS, Rachmilevitch I, et al. Safety and feasibility of multiple blood-brain barrier disruptions for the treatment of glioblastoma in patients undergoing standard adjuvant chemotherapy. J Neurosurg. 2021;134:475–83. - PubMed
Publication types
MeSH terms
Grants and funding
- 92159304, 82372022, 82227806, and 82171958/Natural Science Foundation of China
- 92159304, 82372022, 82227806, and 82171958/Natural Science Foundation of China
- 92159304, 82372022, 82227806, and 82171958/Natural Science Foundation of China
- 92159304, 82372022, 82227806, and 82171958/Natural Science Foundation of China
- no. 2023YFF0714200/National Key Research and Development Program of China
- no. 2023YFF0714200/National Key Research and Development Program of China
- no. 2023YFF0714200/National Key Research and Development Program of China
- no. 2023YFF0714200/National Key Research and Development Program of China
- 2024A1515030212, 2023A1515010747, 2023A1515140133, and 2022A1515140102/Guangdong Basic and Applied Basic Research Fund
- 2024A1515030212, 2023A1515010747, 2023A1515140133, and 2022A1515140102/Guangdong Basic and Applied Basic Research Fund
- 2024A1515030212, 2023A1515010747, 2023A1515140133, and 2022A1515140102/Guangdong Basic and Applied Basic Research Fund
- 2024A1515030212, 2023A1515010747, 2023A1515140133, and 2022A1515140102/Guangdong Basic and Applied Basic Research Fund
- B2302021 and E3A1051001/Shenzhen Medical Research Funds
- B2302021 and E3A1051001/Shenzhen Medical Research Funds
- B2302021 and E3A1051001/Shenzhen Medical Research Funds
- B2302021 and E3A1051001/Shenzhen Medical Research Funds
LinkOut - more resources
Full Text Sources

