Sicca manifestations and lymphoproliferation in hepatitis C virus: effects of direct acting antiviral therapy on dryness and B-cell activity compared to Sjögren's disease
- PMID: 40624706
- PMCID: PMC12232862
- DOI: 10.1186/s13075-025-03605-9
Sicca manifestations and lymphoproliferation in hepatitis C virus: effects of direct acting antiviral therapy on dryness and B-cell activity compared to Sjögren's disease
Abstract
Objectives: Hepatitis C virus (HCV) can be associated with sicca manifestations. To study the effect of direct-acting antivirals (DAAs) on sicca manifestations in HCV-infected patients and the difference between those patients and others with HCV without dryness & Sjögren's disease (SjD).
Methods: We studied 60 patients in 3 groups: Group 1 (20 HCV + sicca), group 2 (20 HCV without sicca), and group 3 (20 SjD). Groups 1 and 2 received DAAs according to the Egyptian Ministry of Health protocols and were evaluated before and after treatment. Group 3 was evaluated once. Our study evaluated the patients by both subjective and objective methods.
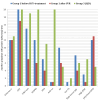
Results: All HCV cases had sustained viral response (SVR). Comparing the characteristics of groups 1 (before treatment) & 3: Group 1 had a higher frequency of RF, cryoglobulins, and polyclonal-hypergammaglobulinemia (P-values 0.021, 0.003, and ˂0.001 respectively). Group 3 had higher scores of VAS dry eye, VAS dry mouth, VAS fatigue, and VAS pain than group 1 (P-values ˂0.001 in all). Group 3 also had a higher frequency of Anti-Ro and Anti-La (P-values < 0.001). Group-1 before DAAs treatment had higher markers denoting B-cell hyperactivity [higher Rheumatoid factor (RF), cryoglobulins, and beta2-microglobulins (β2M)] compared to group-2 which improved markedly after SVR. This supports that group 1 is further ahead in the direction of lymphoproliferation. Group 1 patients after SVR showed marked improvement in VAS dry eye, VAS dry mouth, VAS fatigue, VAS pain, ESSPRI, and ESSDAI (P-values ˂0.003, ˂0.002, ˂0.016, ˂0.001, ˂0.002, and ˂0.014 respectively). There was a significant improvement in RF, and serum β2M levels (after SVR), (P-values ˂0.013, and 0.001 respectively). Group 1 is further ahead in the direction of lymphoproliferation than group 2 with higher statistically significant serum β2M and polyclonal serum protein electrophoresis (P-values 0.006 and 0.047 respectively).
Conclusion: HCV patients with sicca manifestations treated by DAAs showed significant clinical and immunological improvements. The difference between group 1 (before and after SVR) and group 3 supports the notion that they are two different entities, with different characteristic features. Sicca manifestations improved after the eradication of HCV.
Keywords: B-cell proliferation; Direct-acting antiviral drugs; Hepatitis C virus; Sicca manifestations; Sjögren’s disease.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: All procedures performed in the study were in accordance with the ethical standards of the National Research Committee and the 1964 Helsinki Declaration and its later amendments and approved by the Research Ethics Committee, Faculty of Medicine, Cairo University (REC code: D-28-19). A written informed consent was obtained from all participating patients. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures
Similar articles
-
Direct-acting antivirals for chronic hepatitis C.Cochrane Database Syst Rev. 2017 Sep 18;9(9):CD012143. doi: 10.1002/14651858.CD012143.pub3. Cochrane Database Syst Rev. 2017. PMID: 28922704 Free PMC article.
-
Direct-acting antivirals for chronic hepatitis C.Cochrane Database Syst Rev. 2017 Jun 6;6(6):CD012143. doi: 10.1002/14651858.CD012143.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2017 Sep 18;9:CD012143. doi: 10.1002/14651858.CD012143.pub3. PMID: 28585310 Free PMC article. Updated.
-
Pharmacological interventions for acute hepatitis C infection: an attempted network meta-analysis.Cochrane Database Syst Rev. 2017 Mar 13;3(3):CD011644. doi: 10.1002/14651858.CD011644.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2018 Dec 03;12:CD011644. doi: 10.1002/14651858.CD011644.pub3. PMID: 28285495 Free PMC article. Updated.
-
Peginterferon alfa and ribavirin for chronic hepatitis C in patients eligible for shortened treatment, re-treatment or in HCV/HIV co-infection: a systematic review and economic evaluation.Health Technol Assess. 2011 Apr;15(17):i-xii, 1-210. doi: 10.3310/hta15170. Health Technol Assess. 2011. PMID: 21473834 Free PMC article.
-
NIH Consensus Statement on Management of Hepatitis C: 2002.NIH Consens State Sci Statements. 2002 Jun 10-12;19(3):1-46. NIH Consens State Sci Statements. 2002. PMID: 14768714
References
-
- Cacoub P, Buggisch P, Carrión JA, Cooke GS, Zignego AL, Beckerman R et al. Direct medical costs associated with the extrahepatic manifestations of hepatitis C infection in Europe. J Viral Hepat. 2018; 25(7):811-7. 10.1111/jvh.12881, PMID 29476572. - PubMed
-
- Gomes RL, Marques JC, Albers MB, Endo RM, Dantas PE, Felberg S. Ocular surface and hepatitis C. Arquivos Brasileiros De Oftalmologia. 2011;74:97–101. - PubMed
-
- Alves M, Angerami RN, Rocha EM. Dry eye disease caused by viral infection: [review]. Arq Bras Oftalmol. 2013 Mar-Apr; 76(2):129– 32 [review]. 10.1590/s0004-27492013000200016, PMID 23828477. - PubMed
-
- Loustaud-Ratti V, Riche A, Liozon E, Labrousse F, Soria P, Rogez S, et al. Prevalence and characteristics of sjögren’s syndrome or Sicca syndrome in chronic hepatitis C virus infection: a prospective study. J Rheumatol. 2001;28(10):2245–51. PMID 11669164. - PubMed
-
- Carrozzo M. Oral diseases associated with hepatitis C virus infection. Part 1. Sialadenitis and salivary glands lymphoma. Oral Dis. 2008; 14(2):123– 30. 10.1111/j.1601-0825.2007.01436.x, PMID 18208477 [PMID: 18208477 DOI: 10.1111/ j.1601-0825.2007.01436.x]. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

