Activatable theranostic prodrug scaffold with tunable drug release rate for sequential photodynamic and chemotherapy
- PMID: 40625522
- PMCID: PMC12118196
- DOI: 10.1002/smo.20230024
Activatable theranostic prodrug scaffold with tunable drug release rate for sequential photodynamic and chemotherapy
Abstract
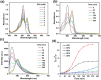
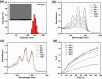
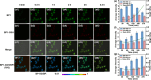
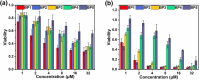
Glutathione (GSH)-activated prodrugs are promising for overcoming the limitations of conventional anti-tumor drugs. However, current GSH-responsive disulfide groups exhibit unregulated reactivity, making it impossible to precisely control the drug release rate. We herein report a series of GSH-responsive prodrugs with a "three-in-one" molecular design by integrating a fluorescence report unit, stimuli-responsive unit and chemodrug into one scaffold with tunable aromatic nucleophilic substitution (SNAr) reactivity. The drug release rate of these prodrugs is tailored by modification of substituent groups with different electron-withdrawing or -donating abilities on the BODIPY core. Furthermore, the prodrugs self-assemble in water to form nanoparticles that serve as photosensitizers to produce reactive oxygen species upon irradiation for photodynamic therapy (PDT). The PDT process also increases the concentration of GSH in cells, further promoting the release of drugs for chemotherapy. This strategy provides a powerful platform for sequential photodynamic and chemotherapy with tunable drug release rates and synergistic therapeutic effects.
Keywords: combinational therapy; fluorescent probes; photodynamic therapy; prodrugs; theranostic agents.
© 2024 The Authors. Smart Molecules published by John Wiley & Sons Australia, Ltd on behalf of Dalian University of Technology.
Conflict of interest statement
There are no conflicts to declare.
Figures

Similar articles
-
Application of photodynamic activation of prodrugs combined with phototherapy in tumor treatment.Mol Cancer. 2025 Jul 21;24(1):200. doi: 10.1186/s12943-025-02404-9. Mol Cancer. 2025. PMID: 40685358 Free PMC article. Review.
-
Interventions for central serous chorioretinopathy: a network meta-analysis.Cochrane Database Syst Rev. 2025 Jun 16;6(6):CD011841. doi: 10.1002/14651858.CD011841.pub3. Cochrane Database Syst Rev. 2025. PMID: 40522203
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of paclitaxel, docetaxel, gemcitabine and vinorelbine in non-small-cell lung cancer.Health Technol Assess. 2001;5(32):1-195. doi: 10.3310/hta5320. Health Technol Assess. 2001. PMID: 12065068
-
Psychological interventions for adults who have sexually offended or are at risk of offending.Cochrane Database Syst Rev. 2012 Dec 12;12(12):CD007507. doi: 10.1002/14651858.CD007507.pub2. Cochrane Database Syst Rev. 2012. PMID: 23235646 Free PMC article.
-
Stimulant and non-stimulant drug therapy for people with attention deficit hyperactivity disorder and epilepsy.Cochrane Database Syst Rev. 2022 Jul 13;7(7):CD013136. doi: 10.1002/14651858.CD013136.pub2. Cochrane Database Syst Rev. 2022. PMID: 35844168 Free PMC article.
References
-
- Chabner B. A., Roberts T. G., Nat. Rev. Cancer 2005, 5, 65. - PubMed
-
- Zhou J., Yu G., Huang F., Chem. Soc. Rev. 2017, 46, 7021. - PubMed
-
- Wu H., Xia T., Qi F., Mei S., Xia Y., Xu J.‐F., Zhang X., CCS Chem. 2023, 5, 823.
-
- Wang P., Jin X., Cai J., Chen J., Ji M., Anti Cancer Agents Med. Chem. 2014, 14, 418. - PubMed
-
- Rautio J., Meanwell N. A., Di L., Hageman M. J., Nat. Rev. Drug Discov. 2018, 17, 559. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
