A spheroid model that recapitulates the protective role of the lymph node microenvironment and serves as a platform for drug testing in chronic lymphocytic leukemia
- PMID: 40625531
- PMCID: PMC12233836
- DOI: 10.1002/hem3.70170
A spheroid model that recapitulates the protective role of the lymph node microenvironment and serves as a platform for drug testing in chronic lymphocytic leukemia
Abstract
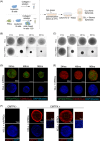
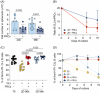
Chronic lymphocytic leukemia (CLL) B cells are characterized by a propensity to undergo rapid apoptosis when cultured in vitro, underscoring the importance of the tissue microenvironment for disease survival. One of the major limitations in studying the role of the microenvironment in tumor development and drug response is the inadequacy of conventional two-dimensional (2D) in vitro assays to physiologically reconstruct the complex spatial organization and interactions of cells in their natural lymphoid niches. To overcome this limitation, we developed a novel in vitro 3D lymph node-like spheroid model of the leukemic microenvironment by culturing human CLL cells with fibroblastic reticular cells (FRCs). FRCs are a key structural component of secondary lymphoid organs and are emerging as crucial players in tissue homeostasis and immune responses. Our results demonstrate that CLL spheroids maintain the physiological cellular ratio between FRCs and leukemic cells over time and protect tumor cells from apoptosis by mimicking the protective effects of the microenvironment. This was further demonstrated by venetoclax treatment that showed reduced apoptosis in 3D compared to a 2D setting. Importantly, the spheroids promote a gene expression profile more aligned with that of CLL cells in lymphoid tissues. The spheroid model provides a straightforward, quick-to-use platform for investigating drug efficacy under conditions that better replicate the natural lymph node microenvironment. This 3D lymph node-like spheroid model could serve as a valuable tool for studying tumor biology and the protective effects of the stromal microenvironment, and for testing therapeutic strategies in a more clinically relevant setting.
© 2025 The Author(s). HemaSphere published by John Wiley & Sons Ltd on behalf of European Hematology Association.
Conflict of interest statement
Paolo Ghia reported the following conflicts of interest: Research funding from AbbVie, AstraZeneca, BMS, and Janssen. Honorarium from AbbVie, AstraZeneca, BeiGene, BMS, Janssen, Loxo@Lilly, MSD, and Roche.
Figures


References
LinkOut - more resources
Full Text Sources
