Recent progress in stimuli-activable metallo-prodrugs for cancer therapy
- PMID: 40625908
- PMCID: PMC12118304
- DOI: 10.1002/smo.20240030
Recent progress in stimuli-activable metallo-prodrugs for cancer therapy
Abstract
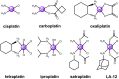
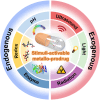
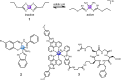
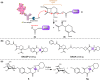
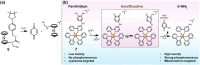
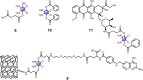
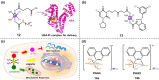
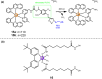
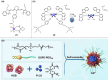
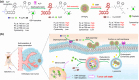
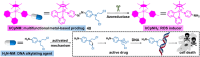
The clinical approval of platinum-based drugs has prompted the development of novel metallo-complexes during the last several decades, while severe problems, especially for poor water solubility, drug resistance and toxicity in patients, greatly hindered the clinical trials and curative efficacy. To address these issues, the concept of metallo-prodrugs has been proposed for oncology. Some stimuli-activable metallo-prodrugs provide new insights for designing and preparing site-specific prodrugs with maximized therapeutic efficacy and negligible unfavorable by-effects. In this review, recent progress in stimuli-activable metallo-prodrugs in the past 20 years has been overviewed, where endogenous and exogenous stimuli have been involved. Typical examples of smart stimuli-activable metallo-prodrugs are discussed regarding to their molecular structure, activation mechanism, and promising biomedical applications. In the end, challenges and future perspectives in metallo-prodrugs have been discussed.
Keywords: cancer therapy; metallo‐prodrugs; stimuli‐activable.
© 2024 The Author(s). Smart Molecules published by John Wiley & Sons Australia, Ltd on behalf of Dalian University of Technology.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





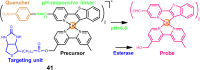
References
-
- a) Mjos K. D., Orvig C., Chem. Rev. 2014, 114, 4540. - PubMed
- b) Gou Y., Huang G., Li J., Yang F., Liang H., Coord. Chem. Rev. 2021, 441, 213975.
-
- a) Wang X., Wang X., Jin S., Muhammad N., Guo Z., Chem. Rev. 2019, 119, 1138. - PubMed
- b) Rilak Simović A., Masnikosa R., Bratsos I., Alessio E., Coord. Chem. Rev. 2019, 398, 113011.
- c) Huang K.‐B., Wang F.‐Y., Lu Y., et al., Proc. Natl. Acad. Sci. U.S.A. 2024, 121, e2404668121. - PMC - PubMed
- d) Lu Y., Wang S.‐S., Li M.‐Y., et al., Acta Pharm. Sin. B 2024. 10.1016/j.apsb.2024.06.017 - DOI
- e) Yang Q.‐Y., Ma R., Gu Y.‐Q., et al., Angew. Chem. Int. Ed. 2022, 61, e202208570. - PubMed
-
- Hartinger C. G., Dyson P. J., Chem. Soc. Rev. 2009, 38, 391. - PubMed
-
- Storr T., Thompson K. H., Orvig C., Chem. Soc. Rev. 2006, 35, 534. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous
