First-in-human phase I open-label study of the anti-TIM-3 monoclonal antibody INCAGN02390 in patients with select advanced or metastatic solid tumors
- PMID: 40631773
- PMCID: PMC12238946
- DOI: 10.1093/oncolo/oyaf144
First-in-human phase I open-label study of the anti-TIM-3 monoclonal antibody INCAGN02390 in patients with select advanced or metastatic solid tumors
Abstract
Background: T-cell immunoglobulin and mucin domain-containing protein-3 (TIM-3) is an immune checkpoint receptor upregulated during anti-programmed death protein-1 (PD-1)/programmed death ligand-1 (PD-L1) immunotherapy for cancer. TIM-3 blockade may improve the antitumor activity of PD-1/PD-L1inhibition. This phase 1 study evaluated INCAGN02390, a novel, fully human Fc-engineered antibody against TIM-3.
Methods: INCAGN02390 was evaluated by dose escalation at 10-1600 mg infused in 14-day cycles (every 2 weeks [Q2W]) in pretreated patients with select advanced/metastatic immunogenic solid tumors. Objectives included evaluation of safety/tolerability and maximum tolerated dose (MTD) (primary), pharmacokinetics, preliminary antitumor activity, pharmacodynamics, and immunogenicity (secondary).
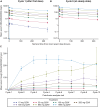
Results: Forty patients were enrolled and treated with INCAGN02390; 60% had previously received ≥3 lines of systemic therapy. Forty-eight percent had received a prior immune checkpoint inhibitor (anti-PD-1/PD-L1 therapy, 43%; anti-cytotoxic T-lymphocyte associated protein-4 therapy, 23%). No dose-limiting toxicities (DLTs) were observed and MTD was not reached. Twelve patients (30%) had treatment-related adverse events (TRAEs), most commonly fatigue and pruritus (n = 3 each); 3 (8%) had grade ≥3 TRAEs. Four patients (10%) experienced sponsor-assessed irAEs. One patient (3%) achieved partial response (duration, 5.7 months) and 6 had stable disease (≥56 days in all patients, >18 months in 2 patients).
Conclusions: In this heavily pretreated population, no DLTs were reported and modest efficacy was exhibited. A 400-mg Q2W dose was selected for phase II studies investigating INCAGN02390 as part of combination immunotherapies for advanced cancers.
Keywords: carcinoma; clinical trial phase I; immune checkpoint inhibitors; immunotherapy; metastases; monoclonal antibodies.
© The Author(s) 2025. Published by Oxford University Press.
Conflict of interest statement
M.E.G. reports stock and other ownership interests from COTA Healthcare; speakers’ bureaus for Bristol Myers Squibb, Eli Lilly & Company, and Merck; research funding from Acerta Pharma, Adlai Nortye, Arcus Biosciences, Array BioPharma, Bayer, Bellicum Pharmaceuticals, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Checkpoint Therapeutics, Compass Therapeutics, Constellation Pharmaceuticals, Cullinan Oncology, Cyteir, Daiichi Sankyo, Eisai, EMD Serono, Erasca, Fate Therapeutics, Georgetown University, GlaxoSmithKline, GSB Pharma, Hackensack Meridian Health, Imugene, Incyte Corporation, Infinity Pharmaceuticals, iTeos Therapeutics, Janssen, Johnson & Johnson, KSQ Therapeutics, MedImmune, Memorial Sloan Kettering Cancer Center, Merck, Millennium Pharmaceuticals, Mirati Therapeutics, Moderna, NextCure, Nimbus Therapeutics, Pfizer, Pharmacyclics, Rapa Therapeutics, Regeneron, Roche/Genentech, Sanofi, Seagen, Silenseed, Synlogic Therapeutics, TESARO, Turning Point Therapeutics, Vedanta Biosciences, VelosBio, Verastem Oncology, and Vincerx Pharma; and travel, accommodations, and expenses from Guardant Health. S.-C.T. reports none. J.D.P. reports being Board of Directors member, with ownership interest and intellectual property, for BioCytics and Carolina BioOncology Institute; founder and owner of BioCytics; founder and owner of a phase 1 cancer research clinic; developing intellectual property for cellular therapies and developing immune cellular therapy as founder/owner of BioCytics; consultant for Aavocyte and Boxer Capital; principal investigator for, without financial interest in, AbbVie, Adagene, Alkermes, Apollo, Apros Therapeutics, Arcus Biosciences, Astellas, AstraZeneca/MedImmune, Atreca, BioNTech, BJ BioScience, Bristol Myers Squibb, Calico Life Sciences, Conjupro BioTherapeutics, Corbus, Cullinan Therapeutics, CytoMx, EMD Serono, Fate Therapeutics, FBD Biologics, FLX Bio, Genentech/Roche, Glenmark, I-MAB Biopharma, Immune-Onc Therapeutics, Incyte, Jounce Therapeutics, MacroGenics, MBrace, Molecular Templates, Multitude, NextCure, Nuvation Bio, Repertoire Immune Medicines, Revolution Medicines, Sairopa, Seattle Genetics, Sequenom, SK Life Sciences, Tempest Therapeutics, Top Alliance Biosciences, Trethera, Xilio Therapeutics, and Zenshine Pharmaceuticals; institutional funding, with no financial interest, from Merck, MT Group, Precision for Medicine, Replimune, STEMCELL Technologies, and Xilis; principal investigator for, with financial interest in, Allarity, Aptevo, Aulos, CUE BioPharma, GI Innovation, Harbour BioMed, IconOVir Bio, IGM BioSciences, Medikine, Moderna TX, PEEL Therapeutics, Phanes Therapeutics, Pieris Pharmaceuticals, Qurgen, RiboScience, Simcere, and SK Life Science; personal and institutional funding from AavoCyte, AbbVie, Adagene, Alkerme, Allarity, Apollo, Apros, Aptevo, Arcus Biosciences, Astellas, AstraZeneca/MedImmune, Atreca, Aulos, BioNTech, BJ BioScience, Bristol Myers Squibb, Calico Life Sciences, Conjupro BioTherapeutics, Corbus, CUE BioPharma, Cullinan, CytoMx, EMD Serono, Fate Therapeutics, FBD Biologics, FLX Bio, Genentech/Roche, GI Innovation, Glenmark, Harbour BioMed, I-MAB Pharma, IconOVIR Bio, IGN BioSciences, Immune-Onc, Incyte, Jounce Therapeutics, Macrogenics, MBrace, Medikine, Moderna TX, Molecular Templates, Multitude, NextCure, Nuvation, PEEL Therapeutics, Phanes Therapeutics, Pieris Pharmaceuticals, PIOMA, Qurgen, Repertoire Immune Medicines, Revolution Medicines, RiboScience, Sairopa, Seattle Genetics, Sequenom, Simcere, SK Life Sciences, Tempest Therapeutics, Top Alliance BioScience, Trethera, Wugen, Xilio Therapeutics, and Zenshine Pharma; funding, with a financial interest, from PIOMA and Wugen; Wugen is sponsor of contract laboratory translational research; writing engagement, personal, and other, with financial interest, from Aaavocyte; AavoCyte & AavoBioCytics are jointly developing cellular therapies with BioCytics Human Applications Lab for point-of-care manufacturingother; personal, from Boxer Capital; clinical trial funding from Apollo, Astellas, BioNTech, Corbus, CytoMx, FBD Biologics, Glenmark, MBrace, Multitude, Revolution Medicines, Sairopa, and SK Life Sciences; and funding for contract laboratory services from Nuvation. A.S.B. reports speakers’ bureaus for AstraZeneca, Bristol Myers Squibb, Genentech, Merati, Merck, and Regeneron; steering committee for Janssen (Johnson & Johnson) (2023, 2024); and consultant for AbbVie (2024) and Pfizer (2023). P.E.H., Z.D., L.C., X.G., and J.E.J., report employment by and stock ownership in Incyte Corporation. N.B. reports employment by and stock ownership in Incyte Biosciences International Sàrl. O.H. reports honoraria from Bristol Myers Squibb, Immunocore, Novartis, Pfizer, and Regeneron; has served as a consultant or advisor to Alkermes, Amgen, Bactonix, BeiGene, BioAtla, Bristol Myers Squibb, Eisai, Georgiamune, GigaGen, GlaxoSmithKline, Grit Biotechnology, Idera, Immunocore, Incyte Corporation, Instil Bio, Iovance Biotherapeutics, Janssen, KSQ Therapeutics, Merck, Moderna, Novartis, Obsidian, Pfizer, Regeneron, Roche/Genentech, Sanofi, Seattle Genetics, Tempus, Vial Health, and Zelluna Immunotherapy; has participated in speakers’ bureaus for Bristol Myers Squibb, Immunocore, Novartis, Pfizer, and Regeneron; has received institutional research funding from Arcus Biosciences, Aduro, Akeso, Amgen, Bio Atla, Bristol Myers Squibb, CytomX Therapeutics, Exelixis, GlaxoSmithKline, Idera, Immunocore, Incyte Corporation, Iovance Biotherapeutics, Merck, Merck Serono, Moderna, NextCure, Novartis, Pfizer, Regeneron, Roche/Genentech, Seattle Genetics, Torque Pharmaceuticals, and Zelluna Immunotherapy.
Figures

References
-
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. https://doi.org/ 10.1038/nrc3239 - DOI - PMC - PubMed
-
- Yuan Y, Adam A, Zhao C, Chen H.. Recent advancements in the mechanisms underlying resistance to PD-1/PD-L1 blockade immunotherapy. Cancers (Basel). 2021;13:663. https://doi.org/ 10.3390/cancers13040663 - DOI - PMC - PubMed
-
- Brahmer JR, Abu-Sbeih H, Ascierto PA, et al. Society for Immunotherapy of Cancer (SITC) clinical practice guideline on immune checkpoint inhibitor-related adverse events. J ImmunoTher Cancer. 2021;9:e002435. https://doi.org/ 10.1136/jitc-2021-002435 - DOI - PMC - PubMed
-
- Meng L, Wu H, Wu J, et al. Mechanisms of immune checkpoint inhibitors: insights into the regulation of circular RNAS involved in cancer hallmarks. Cell Death Dis. 2024;15:3. https://doi.org/ 10.1038/s41419-023-06389-5 - DOI - PMC - PubMed
-
- Sun J-Y, Zhang D, Wu S, et al. Resistance to PD-1/PD-L1 blockade cancer immunotherapy: mechanisms, predictive factors, and future perspectives. Biomark Res. 2020;8:35. https://doi.org/ 10.1186/s40364-020-00212-5 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

