Feasibility and preliminary efficacy of heavy Lifting Strength Training versus Usual Care in Head and Neck Cancer Survivors (the LIFTING 2 Trial): A study protocol for a single-centre, phase II, randomized controlled trial
- PMID: 40638621
- PMCID: PMC12244484
- DOI: 10.1371/journal.pone.0326860
Feasibility and preliminary efficacy of heavy Lifting Strength Training versus Usual Care in Head and Neck Cancer Survivors (the LIFTING 2 Trial): A study protocol for a single-centre, phase II, randomized controlled trial
Abstract
Background: Despite improvements in treatments, head and neck cancer survivors (HNCS) still endure acute and chronic side effects such as loss of muscular strength, limitations in physical function, fatigue, and swallowing difficulties that impact quality of life (QoL). Light-to-moderate intensity strength training (LMST) has been shown to improve some of these side effects. Heavy lifting strength training (HLST) may further improve outcomes, however, only one pilot study has focused on HNCS. The primary aim of this study is to further establish the feasibility of HLST in HNCS. A secondary aim is to provide preliminary evidence of the effects of a HLST program compared to Usual Care (UC) in HNCS.
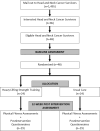
Methods: This single-centre, two-armed, randomized controlled trial will aim to recruit 48 HNCS ≥1-year post-treatment, and randomly assign them to a 12-week Heavy Lifting Strength Training (HLST) group or Usual Care (UC) group. With 80% power, a two-tailed alpha of p < 0.05, and adjusting for covariates that explain 25% of the variance in the outcome, we will be able to detect a standardized effect size of 0.80 with 38 (19/group) evaluable HNCS, allowing for a 20% loss to follow-up. The HLST group will exercise twice weekly, progressing to lifting low repetitions of heavy loads at 80% to 100% of maximal perceived exertion, whereas the UC group will not receive any exercise prescription or instruction during the 12-week intervention. The primary efficacy outcome will be upper and lower body muscular strength assessed by 3RM tests. Secondary efficacy outcomes will include health-related fitness and patient-reported outcomes assessed with reliable physical assessments and validated questionnaires.
Significance: Our findings will answer the important question of whether lifting heavier weights is feasible for HNCS and leads to better outcomes compared to lifting no weights. The results will inform future studies comparing exercise intensities in HNCS and other cancer populations.
Copyright: © 2025 Ntoukas et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
Physical exercise training interventions for children and young adults during and after treatment for childhood cancer.Cochrane Database Syst Rev. 2016 Mar 31;3(3):CD008796. doi: 10.1002/14651858.CD008796.pub3. Cochrane Database Syst Rev. 2016. PMID: 27030386 Free PMC article.
-
Physical exercise training interventions for children and young adults during and after treatment for childhood cancer.Cochrane Database Syst Rev. 2013 Apr 30;(4):CD008796. doi: 10.1002/14651858.CD008796.pub2. Cochrane Database Syst Rev. 2013. Update in: Cochrane Database Syst Rev. 2016 Mar 31;3:CD008796. doi: 10.1002/14651858.CD008796.pub3. PMID: 23633361 Updated.
-
Corticosteroids for the treatment of Duchenne muscular dystrophy.Cochrane Database Syst Rev. 2016 May 5;2016(5):CD003725. doi: 10.1002/14651858.CD003725.pub4. Cochrane Database Syst Rev. 2016. PMID: 27149418 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical