Modeling antibody persistence after MenACYW-TT vaccination and comparative analysis with other quadrivalent meningococcal vaccines
- PMID: 40640249
- PMCID: PMC12246452
- DOI: 10.1038/s41598-025-08112-0
Modeling antibody persistence after MenACYW-TT vaccination and comparative analysis with other quadrivalent meningococcal vaccines
Abstract
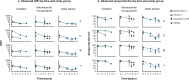
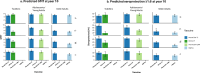
Evaluating the persistence of antibody titers produced by quadrivalent meningococcal vaccines is crucial for determining the optimal timing for primary and booster doses. Using 3 - 7-year persistence data after a single priming dose of MenACYW-TT to fit a statistical model of antibody decay over time (10 years), this analysis modeled long-term antibody persistence for this vaccine in toddlers (12-23 months), adolescents/young adults (13-26 years), and older adults (≥ 56 years), comparing it with other vaccines (MCV4-TT, MenACWY-CRM, MPSV4). The statistical model is based on a Bayesian approach and it accounts for vaccine- and age group-specific antibody decline, missing data, assay errors, and antibody boosting from breakthrough infections. At 10 years post-vaccination, it predicted comparable or higher seroprotective immunopersistence for MenACYW-TT (titers ≥ 1:8 with hSBA) versus (1) MCV4-TT in toddlers (77% [95% CI 70-84] vs. 17% [6-31] for serogroup C, 67% [59-74] vs. 36% [20-53] for serogroup W); (2) MenACWY-CRM in adolescents/young adults (63% [55-71] vs. 40% [32-48] for serogroup C, 67% [59-74] vs. 57% [47-67] for serogroup W); and (3) MPSV4 in older adults (31% [23-39] vs. 22% [14-29] for serogroup C, 38% [31-46] vs. 20% [14-27] for serogroup W). In conclusion, our analysis indicated similar or higher immune persistence at 10 years for MenACYW-TT compared with other quadrivalent meningococcal vaccines, particularly for serogroups C and W.
Keywords: Invasive meningococcal disease; Long-term immunopersistence; MenACYW-TT; Modelling; Quadrivalent meningococcal vaccines; Serogroups A, C, W, Y; Seroprotection.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: LC, IB-G, CZ, EL, SB, FC and PO are employees of Sanofi and may hold shares and/or stock options in the company. Ethical approval: This study was conducted in accordance with the protocol and consensus ethical principles derived from international guidelines including the Declaration of Helsinki, and the International Council for Harmonisation (ICH) guidelines for Good Clinical Practice (GCP), all applicable laws, rules, and regulations.
Figures


References
-
- Centers for Disease Control and Prevention. Clinical Overview of Meningococcal Disease, https://www.cdc.gov/meningococcal/hcp/clinical/index.html?CDC_AA_refVal=... (2024).
-
- Centers for Disease Control and Prevention. Enhanced Meningococcal Disease Surveillance Report, 2022: Confirmed and Probable Cases Reported to the National Notifiable Diseases Surveillance System, https://www.cdc.gov/meningococcal/downloads/NCIRD-EMS-Report-2022-508.pdf (2024).
-
- Booy, R., Gentile, A., Nissen, M., Whelan, J. & Abitbol, V. Recent changes in the epidemiology of Neisseria meningitidis serogroup W across the world, current vaccination policy choices and possible future strategies. Hum. Vaccine Immunother.15, 470–480. 10.1080/21645515.2018.1532248 (2019). - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

