Evaluation of PCV2 vaccine immunogenicity and efficacy using ELISpot to detect virus-specific memory B cells
- PMID: 40640918
- PMCID: PMC12247238
- DOI: 10.1186/s40813-025-00452-7
Evaluation of PCV2 vaccine immunogenicity and efficacy using ELISpot to detect virus-specific memory B cells
Abstract
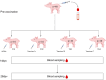
Background: Porcine Circovirus 2 (PCV2) vaccination plays a crucial role in preventing porcine circovirus-associated disease (PCVAD). Nevertheless, pig farms face significant challenges in evaluating vaccination efficacy due to the inability of PCV2 vaccines to achieve sterilizing immunity and the variability among vaccine manufacturers. These challenges are further compounded by the limitations of conventional antibody detection methods, which fail to distinguish between maternally-derived antibodies (MDAs) and vaccine-induced antibodies. The accurate evaluation and selection of PCV2 vaccines is critical for the swine industry. The present study aimed to develop an Enzyme-linked immunospot (ELISpot) assay for directly detecting PCV2-specific memory B cells. This approach was used to assess the presence of PCV2-specific memory B cells in piglets with high levels of MDA vaccinated with different PCV2 vaccines, thus enabling the evaluation of vaccine immunogenicity at the cellular level. Furthermore, antibody levels and the viremia status were analyzed using Enzyme-linked immunosorbent assay (ELISA) and quantitative real-time polymerase chain reaction (qPCR) respectively to provide a comprehensive assessment of the ELISpot assay potential for evaluating the vaccine immunogenicity of PCV2 vaccines.
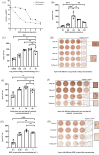
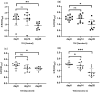
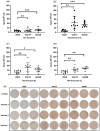
Results: The findings revealed that the optimal conditions for the developed ELISpot assay included stimulation with R848 at a final concentration of 1 µg·mL⁻¹ for three days, a PCV2 Cap protein coating concentration of 1.25 µg·mL⁻¹, a biotinylated goat anti-pig IgG antibody concentration of 5 µg·mL⁻¹, and an HRP-streptavidin concentration of 0.25 µg·mL⁻¹. In high MDA piglets immunized with different vaccines, serum antibody detection showed that PCV2 antibody levels declined continuously over time in all vaccinated and saline-injected control groups, demonstrating similar trends. In contrast, ELISpot analysis demonstrated a significant increase in PCV2-specific memory B cell levels in all three vaccinated groups compared to the saline-injected group. Among the vaccines tested, Vaccine A induced the highest levels of specific memory B cells, followed by Vaccine B. This was consistent with the lower PCV2 infection rates and viremia levels observed in Vaccine A and Vaccine B groups, compared to Vaccine C and saline-injected control groups.
Conclusions: We established an ELISpot assay to quantify PCV2-specific memory B cells, revealing that vaccinated piglets with high MDA levels developed robust memory B cell responses. However, levels of PCV2 IgG antibodies in vaccinated piglets remained statistically indistinguishable from control piglets. These findings demonstrate that ELISpot-based profiling of PCV2-specific memory B cells overcomes the confounding effects of MDA in vaccine efficacy assessments. This approach reliably reflects the humoral immune response induced by vaccination and its relevance in combating natural PCV2 infection, providing valuable guidance for preventing and controlling PCVAD.
Keywords: ELISpot; Immune memory; Memory B cells; Porcine circovirus 2; Vaccine.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval: The animal study was reviewed and approved by the Animal Experiments Ethics Committee of Hunan Agricultural University. The Experimental Animal Care and Use Guidelines of the Ministry of Science and Technology of China (MOST-2011-02) were strictly followed. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures
Similar articles
-
Immunogenicity and seroefficacy of pneumococcal conjugate vaccines: a systematic review and network meta-analysis.Health Technol Assess. 2024 Jul;28(34):1-109. doi: 10.3310/YWHA3079. Health Technol Assess. 2024. PMID: 39046101 Free PMC article.
-
Antibody tests for identification of current and past infection with SARS-CoV-2.Cochrane Database Syst Rev. 2022 Nov 17;11(11):CD013652. doi: 10.1002/14651858.CD013652.pub2. Cochrane Database Syst Rev. 2022. PMID: 36394900 Free PMC article.
-
Development a high-sensitivity sandwich ELISA for determining antigen content of porcine circovirus type 2 vaccines.J Virol Methods. 2024 Jul;328:114954. doi: 10.1016/j.jviromet.2024.114954. Epub 2024 May 17. J Virol Methods. 2024. PMID: 38763359
-
Efficacy of PCV2 Vaccination Under Natural Conditions: A Longitudinal Study Using PCR and Virus Isolation.Vet Sci. 2025 Jun 11;12(6):575. doi: 10.3390/vetsci12060575. Vet Sci. 2025. PMID: 40559812 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
References
-
- Allan GM, McNeilly F, Kennedy S, Daft B, Clarke EG, Ellis JA, et al. Isolation of Porcine circovirus-like viruses from pigs with a wasting disease in the USA and Europe. J Vet Diagn Investig Off Publ Am Assoc Vet Lab Diagn Inc. 1998;10:3–10. - PubMed
-
- Wang Y, Noll L, Lu N, Porter E, Stoy C, Zheng W, et al. Genetic diversity and prevalence of Porcine circovirus type 3 (PCV3) and type 2 (PCV2) in the Midwest of the USA during 2016–2018. Transbound Emerg Dis. 2020;67:1284–94. - PubMed
-
- Zhang D, He K, Wen L, Fan H. Genetic and phylogenetic analysis of a new Porcine circovirus type 2 (PCV2) strain in China. Arch Virol. 2015;160:3149–51. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

