Characteristics, preparation and applicability in oral delivery systems of cellulose ether-based buccal films
- PMID: 40641388
- PMCID: PMC12258245
- DOI: 10.1080/10717544.2025.2525223
Characteristics, preparation and applicability in oral delivery systems of cellulose ether-based buccal films
Abstract
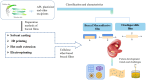
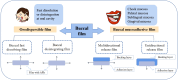
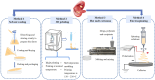
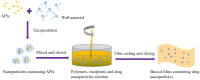
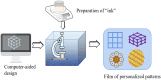
With the rapid development of buccal films (BFs), the demand for film-forming materials and preparation techniques has increased. Cellulose ethers (CEs) exhibit favorable properties, such as effective film formation, mucosal adhesion, and biocompatibility; as such, they are most commonly employed as film-forming materials, essential for BF fabrication. CE-based BFs, classified as orodispersible and buccal mucoadhesive films, can be prepared through solvent casting, inkjet printing, three-dimensional printing, electrospinning, and hot melt extrusion. Hydrophilic CE-based orodispersible films can rapidly dissolve or disintegrate upon contact with saliva to release drugs. High-viscosity or hydrophobic CEs can serve as protective layers for BFs, controlling the unidirectional release of drugs and mitigating the effects of saliva and buccal movements. These mucoadhesive films can firmly adhere to the buccal mucosa for an extended period, prolonging drug release time and enhancing bioavailability. CEs come in various types and grades, exhibiting different rheological and physicomechanical properties, which also provide options for customized design to specific patients. This review provides an overview of CE-based BF technology, analyzes the challenges and development directions of this film, and identifies key areas for scientific research, such as the interactions of bioadhesive materials in buccal mucosal drug delivery. The objectives of this review are to (i) highlight the value of their application in oral drug delivery and (ii) promote the broader adoption of BF-based patient-centric dosing.
Keywords: Cellulose ether; buccal film; drug delivery system; mucoadhesion; preparation method.
Conflict of interest statement
The authors declare that they have no competing financial interests or personal relationships that could have influenced the work reported in this manuscript.
Figures


Similar articles
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
Mucoadhesive in situ forming Gels: Bridging Formulation design and physiological Realities.Int J Pharm. 2025 Aug 20;681:125834. doi: 10.1016/j.ijpharm.2025.125834. Epub 2025 Jun 9. Int J Pharm. 2025. PMID: 40499601
-
Management of urinary stones by experts in stone disease (ESD 2025).Arch Ital Urol Androl. 2025 Jun 30;97(2):14085. doi: 10.4081/aiua.2025.14085. Epub 2025 Jun 30. Arch Ital Urol Androl. 2025. PMID: 40583613 Review.
-
Sexual Harassment and Prevention Training.2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 36508513 Free Books & Documents.
References
-
- Abo-Shady AZ, Elkammar H, Elwazzan VS, Nasr M.. 2020. Formulation and clinical evaluation of mucoadhesive buccal films containing hyaluronic acid for treatment of aphthous ulcer. J Drug Delivery Sci Technol. 55(C):101442. doi: 10.1016/j.jddst.2019.101442. - DOI
-
- Abouhussein D, El Nabarawi MA, Shalaby SH, El-Bary AA.. 2020. Cetylpyridinium chloride chitosan blended mucoadhesive buccal films for treatment of pediatric oral diseases. J Drug Delivery Sci Technol. 57:101676. doi: 10.1016/j.jddst.2020.101676. - DOI
-
- Aldawsari HM, Badr-Eldin SM.. 2020. Enhanced pharmacokinetic performance of dapoxetine hydrochloride via the formulation of instantly-dissolving buccal films with acidic pH modifier and hydrophilic cyclodextrin: factorial analysis, in vitro and in vivo assessment. J Adv Res. 24:281–290. doi: 10.1016/j.jare.2020.04.019. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
