Uptakes of boronophenylalanine in the in vitro and in situ glioblastoma and the potential efficacy of boron neutron capture therapy
- PMID: 40642292
- PMCID: PMC12241995
- DOI: 10.1016/j.ibneur.2025.06.014
Uptakes of boronophenylalanine in the in vitro and in situ glioblastoma and the potential efficacy of boron neutron capture therapy
Abstract
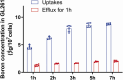
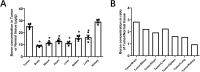
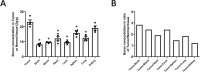
Human glioblastoma (GBM) is the most malignant brain tumor. Boron neutron capture therapy (BNCT) is proved to be a new technology for the effective treatment of GBM. We previously reported that boronophenylalanine (BPA), the boron drug used in BNCT prefers to the tumor of a mouse subcutaneous tumor model injected U87 and GL261, two GBM cell lines. The present study was designed to investigate the distribution of BPA in the tumor of a mouse in situ brain tumor model injected U87 and GL261 in the brain. The tumor model was evaluated by using small animal magnetic resonance imaging (MRI) and confirmed with the morphological observations. The boron concentration indicative of BPA in cells and tumor was measured using inductively coupled plasma atomic emission spectroscopy (ICP-AES). The results revealed that the protein of L-type amino acid transporter (LAT1) was highly expressed in both U87 and GL261 cells. In addition, boron concentration in U87 and GL261 cells was increased 1, 2, 3, 5 and 7 h after addition of BPA in a time-dependently manner. However, the boron concentration in the cells was rapidly decreased when the BPA in the medium was withdrawn for 1 h at each time point, reaching almost the same levels between the time points. In one mouse in situ brain tumor model injected U87 cells, the concentration of boron in the tumor tissue (25.529 μg/g) was higher than that in brain tissue (8.973 μg/g), blood (11.407 μg/g), heart (13.131 μg/g), liver (11.271 μg/g), spleen (15.631 μg/g) and lung (16.226 μg/g) respectively, having ratios of tumor/normal tissue 2.845, 2.238, 1.944, 2.265, 1.633 and 1.537 respectively. Similarly, in another mouse in situ brain tumor model injected GL261 cells, the concentration of boron in the tumor tissue (23.039 μg/g) was higher than that in brain tissue (8.141 μg/g), blood (9.573 μg/g), heart (12.119 μg/g), liver (9.609 μg/g), spleen (15.852 μg/g) and lung (12.565 μg/g) respectively, having ratios of tumor/normal tissue 2.831, 2.407, 1.901, 2.398, 1.453 and 1.834 respectively. The results suggest that uptakes of BPA in two cell lines occur in a time-dependent manner and that BPA preferred to distribute in brain tumor tissue than other normal tissues, having potential efficacy of BNCT.
Keywords: Boron distribution; Boron neutron capture therapy; Boronophenylalanine; Brain tumor cell lines; In situ brain tumor model.
© 2025 The Authors.
Conflict of interest statement
All authors have nothing to declare.
Figures


References
-
- Barth R.F., Carpenter D.E. Rodent brain tumor models for studies focusing on boron neutron capture therapy. Cancer Biother. Radiopharm. 2023;38:148–151. - PubMed
-
- Chen Y.W., Lee Y.Y., Lin C.F., Pan P.S., Chen J.K., Wang C.W., Hsu S.M., Kuo Y.C., Lan T.L., Hsu S.P.C., Liang M.L., Chen R.H., Chang F.C., Wu C.C., Lin S.C., Liang H.K., Lee J.C., Chen S.K., Liu H.M., Peir J.J., Lin K.H., Huang W.S., Chen K.H., Kang Y.M., Liou S.C., Wang C.C., Pai P.C., Li C.W., Chiek D.Q.S., Wong T.T., Chiou S.H., Chao Y., Tanaka H., Chou F.I., Ono K. Salvage boron neutron capture therapy for malignant brain tumor patients in compliance with emergency and compassionate use: evaluation of 34 cases in Taiwan. Biology (Basel) 2021;10 - PMC - PubMed
-
- Kageji T., Mizobuchi Y., Nagahiro S., Nakagawa Y., Kumada H. Clinical results of boron neutron capture therapy (BNCT) for glioblastoma. Appl. Radiat. Isot. 2011;69:1823–1825. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
