The Cholesterol Biosynthesis Pathway Plays an Important Role in Chemotherapeutic Drug Response and Metastasis in High-Grade Osteosarcoma
- PMID: 40643514
- PMCID: PMC12249065
- DOI: 10.3390/cells14130993
The Cholesterol Biosynthesis Pathway Plays an Important Role in Chemotherapeutic Drug Response and Metastasis in High-Grade Osteosarcoma
Abstract
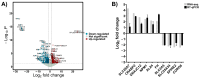
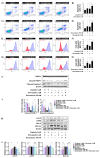
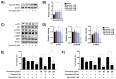
High-grade osteosarcoma (HGOS) is the most common primary malignant bone tumor in children and adolescents. Poor response to chemotherapy is linked to worse prognosis and increased risk of recurrence and metastasis. However, current assessment methods, such as tumor necrosis evaluation, are time-consuming and delay treatment decisions. Thus, identifying molecular pathways and predictive biomarkers is essential for guiding early therapeutic strategies. In this study, RNA-seq analysis of HGOS tissues revealed enrichment of cholesterol biosynthesis and mitotic pathways in poor responders. Additionally, high HMGCR expression, as analyzed from TCGA data, was associated with poor prognosis in sarcoma. Functional validation using SaOS-2 cells, which exhibited poor drug sensitivity and elevated HMGCR levels, demonstrated that simvastatin enhanced the efficacy of cisplatin and doxorubicin by inducing mitochondrial-mediated apoptosis and downregulating anti-apoptotic proteins. Simvastatin also reduced cell migration and invasion by suppressing epithelial-mesenchymal transition and extracellular matrix degradation. Mechanistically, simvastatin disrupted Ras prenylation and inhibited downstream oncogenic signaling pathways, including Akt/mTOR and Akt/GSK3, which regulate survival and metastasis-associated gene expression. These findings suggest that the cholesterol biosynthesis pathway particularly plays a critical role in chemoresistance and metastasis in HGOS and may serve as a promising predictive molecular target for guiding early therapeutic strategies.
Keywords: RNA-seq; cholesterol biosynthesis; drug resistance; osteosarcoma; transcriptomics.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of the data; in the writing of the manuscript; or in the decision to publish the results.
Figures



Similar articles
-
Inhibition of circ_0127646 Enhanced the Cisplatin Sensitivity in Osteosarcoma Cells via miR-22/KAT6B Axis.J Biochem Mol Toxicol. 2025 Jul;39(7):e70381. doi: 10.1002/jbt.70381. J Biochem Mol Toxicol. 2025. PMID: 40557815
-
The EphA2 Receptor Regulates Invasiveness and Drug Sensitivity in Canine and Human Osteosarcoma Cells.Cells. 2024 Jul 16;13(14):1201. doi: 10.3390/cells13141201. Cells. 2024. PMID: 39056783 Free PMC article.
-
Rab27a+CAF exosomal miR-9-5p promotes osteosarcoma progression via CREBRF/MAPK signaling pathway.Cell Signal. 2025 Oct;134:111964. doi: 10.1016/j.cellsig.2025.111964. Epub 2025 Jun 26. Cell Signal. 2025. PMID: 40581265
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of paclitaxel, docetaxel, gemcitabine and vinorelbine in non-small-cell lung cancer.Health Technol Assess. 2001;5(32):1-195. doi: 10.3310/hta5320. Health Technol Assess. 2001. PMID: 12065068
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of topotecan for ovarian cancer.Health Technol Assess. 2001;5(28):1-110. doi: 10.3310/hta5280. Health Technol Assess. 2001. PMID: 11701100
References
-
- Ottaviani G., Jaffe N. The epidemiology of osteosarcoma. Pediatr. Adolesc. Osteosarcoma. 2009;152:3–13. - PubMed
-
- Sever N., Şimşek F., Onur İ.D., Arvas H., Guliyev T., Şakalar T., Çiçek C.M., Orman S., Çetin E.B., Kayaş K., et al. Prognostic Factors in High Grade Osteosarcoma Patients Who Received Neoadjuvant Therapy and Subsequently Underwent Surgery: Data from the Turkish Oncology Group. J. Clin. Med. 2025;14:2024. doi: 10.3390/jcm14062024. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

