Activation of sclerostin inhibits Isg20-Mediated aerobic glycolysis ameliorating renal Fibrosis: the renoprotective mechanism of hederagenin in CKD
- PMID: 40644925
- PMCID: PMC12275953
- DOI: 10.1016/j.redox.2025.103762
Activation of sclerostin inhibits Isg20-Mediated aerobic glycolysis ameliorating renal Fibrosis: the renoprotective mechanism of hederagenin in CKD
Abstract
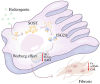
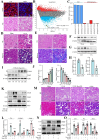
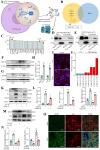
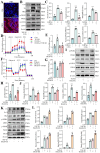
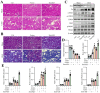
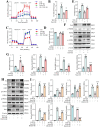
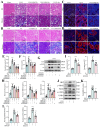
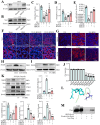
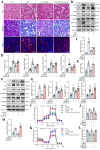
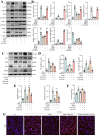
Sclerostin (Sost) functions as an inhibitor of the Wnt/β-catenin signaling pathway, which is known to promote kidney cell epithelial-to-mesenchymal transition (EMT), and fibrosis in chronic kidney disease (CKD). However, the renoprotective effects of Sost in kidney diseases and its therapeutic potential as a target remain unknown. To clarify the protective role of Sost in CKD kidneys, we utilized ultrasound microbubble-mediated renal in situ gene transfection to overexpress and knockdown Sost in kidney. Subsequently, we employed the TurboID-based protein interaction technique to screen for Sost-binding proteins and combined it with ECAR/OCR to elucidate the regulation of glycolytic pathways by Sost and its binding proteins. Sost is predominantly expressed in renal tubules and that its expression is significantly diminished in renal tissues of CKD patients, as well as in UUO and folic acid (FA) induced CKD mouse. Overexpression of Sost in vivo and in vitro ameliorated tubular injury and fibrosis. Employing the TurboID technique, we uncovered an interaction between Sost and the potential glycolysis-associated protein Isg20, an interferon-stimulated ribonuclease. This binding interaction serves to inhibit Isg20-mediated aerobic glycolysis and subsequent fibrosis within the kidney of CKD. For Sost agonists screening, we found that HDG exerts salient anti-fibrotic and renal protective effects in CKD, which are likely attributable to its significant upregulation of Sost expression, thereby inhibiting Isg20-mediated glycolysis. In summary, we demonstrate that upregulation of Sost by HDG inhibits glycolysis and renal fibrosis in CKD through binding and suppressing of Isg20, and targeting Sost may develop novel approaches to treat CKD.
Keywords: CKD; Fibrosis; Hederagenin; Isg20; Sost.
Copyright © 2025 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors have declared no conflict of interest.
Figures
References
-
- Cockwell P., Fisher L.A. The global burden of chronic kidney disease. Lancet. 2020;395:662–664. - PubMed
-
- Li L., Fu H., Liu Y. The fibrogenic niche in kidney fibrosis: components and mechanisms. Nat. Rev. Nephrol. 2022;18:545–557. - PubMed
-
- Liu Y. Kidney fibrosis: fundamental questions, challenges, and perspectives. Integrative Medicine in Nephrology and Andrology. 2024;11:e24–e27.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

