Effect of Pre-IVM Duration with cAMP Modulators on the Production of Cloned Equine Embryos and Foals
- PMID: 40646860
- PMCID: PMC12248448
- DOI: 10.3390/ani15131961
Effect of Pre-IVM Duration with cAMP Modulators on the Production of Cloned Equine Embryos and Foals
Abstract
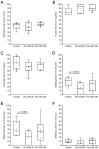
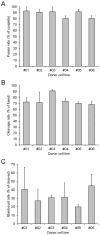
The asynchrony of cytoplasmic and nuclear maturation in cumulus-oocyte complexes (COCs) due to prematurely declining concentrations of cyclic adenosine monophosphate (cAMP) has been shown to result in reduced oocyte developmental competence. The objective of this study was to evaluate the effect of pre-IVM treatment with cAMP modulators for different durations on the developmental potential of equine oocytes used for cloned embryo production. Collected COCs were transferred to cryovials filled with transport medium at 20-22 °C. Within the cryovials, the COCs were either untreated (Control) for 18 h or treated with 50 µM forskolin and 100 µM 3-isobutyl-1-methylxanthine for the first 4 h (Pre-IVM 4 h) or the entire 18 h (Pre-IVM 18 h). Oocytes were then transferred to maturation medium and incubated for a further 22-24 h at 38.5 °C in 5% CO2 in air. Somatic cell nuclear transfer embryos were then produced using the meiotically mature oocytes and donor cells from six different fibroblast cell lines. The rates of maturation and embryo development did not differ significantly between the groups, though blastocyst formation tended to be inferior in the Pre-IVM 4 h group compared with the Control group (p = 0.06). Of 67 blastocysts produced, 23 were transferred to recipient mares on Day 4 or 5 post-ovulation. Regarding the pregnancy outcomes, no significant differences were found between the groups, and four viable foals were born, each derived from a different donor cell line. The findings expand on those from previous evaluations of this biphasic IVM system, and indicate that the cAMP-modulating treatments exert limited effects under the pre-IVM conditions used here.
Keywords: biphasic IVM; blastocyst; embryo transfer; horse; in vitro maturation (IVM); simulated physiological oocyte maturation (SPOM); somatic cell nuclear transfer (SCNT).
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
-
- Galli C., Colleoni S., Duchi R., Lagutina I., Lazzari G. Developmental competence of equine oocytes and embryos obtained by in vitro procedures ranging from in vitro maturation and ICSI to embryo culture, cryopreservation and somatic cell nuclear transfer. Anim. Reprod. Sci. 2007;98:39–55. doi: 10.1016/j.anireprosci.2006.10.011. - DOI - PubMed
LinkOut - more resources
Full Text Sources

