Establishment and Partial Characterization of Canine Mammary Tumor Cell Lines
- PMID: 40646890
- PMCID: PMC12249022
- DOI: 10.3390/ani15131991
Establishment and Partial Characterization of Canine Mammary Tumor Cell Lines
Abstract
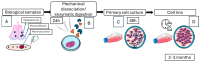
Mammary tumors are the most common neoplasms diagnosed in female dogs and have been considered excellent models for studying human breast cancer. Establishing cell lines from primary cultures of canine mammary tumors provides an in vitro model to better understand the disease and develop new treatments. This study aimed to establish and characterize canine mammary tumor cell lines. Ten cell cultures were generated from tumor tissue obtained from affected dogs, including seven from primary mammary tumors and three from metastatic sites. Characterization included molecular marker expression (ER, PR, HER2, cytokeratin 5/6 (CK5/6), vimentin, and the marker of cell proliferation Ki67) and in vitro tumorigenic capacity assessment. Additionally, the susceptibility of five cell lines to DOX, 5-FU, paclitaxel, colchicine, and carboplatin was evaluated using the MTT assay. ICC analysis revealed negative expression of hormonal receptors (ER and PR) in five cell lines, while only one cell line was positive for both. Six cell lines were HER2-negative and positive for vimentin. Five cell lines exhibited in vitro tumorigenic capacity, forming colonies in soft agar. DOX showed the highest growth-inhibitory effect (DOX > Paclitaxel > Colchicine > 5-FU > Carboplatin). Two cell lines had a minimal concentration for 50% inhibition in vitro (IC50) < 0.63 µM and 4.37 ± 0.40 µM for DOX, while one was sensitive to colchicine and paclitaxel (IC50 0.19 µM and 0.04 µM, respectively). All tested cell lines were resistant to carboplatin and 5-FU. These cell lines provide a valuable model for studying breast cancer in humans and dogs and evaluating new potential therapeutic strategies.
Keywords: breast cancer; cancer; canine mammary tumors; cell culture; cell lines.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Cost-effectiveness of using prognostic information to select women with breast cancer for adjuvant systemic therapy.Health Technol Assess. 2006 Sep;10(34):iii-iv, ix-xi, 1-204. doi: 10.3310/hta10340. Health Technol Assess. 2006. PMID: 16959170
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of topotecan for ovarian cancer.Health Technol Assess. 2001;5(28):1-110. doi: 10.3310/hta5280. Health Technol Assess. 2001. PMID: 11701100
-
Establishment and molecular characterization of novel luminal A and luminal B canine mammary cancer cell lines for comparative oncology.Vet World. 2025 Jun;18(6):1725-1740. doi: 10.14202/vetworld.2025.1725-1740. Epub 2025 Jun 26. Vet World. 2025. PMID: 40689167 Free PMC article.
-
Carboplatin and paclitaxel induced-gonadotoxicity on the ovarian reserve of young breast cancer patients with BRCA1 mutation.Hum Reprod. 2025 Jul 11:deaf133. doi: 10.1093/humrep/deaf133. Online ahead of print. Hum Reprod. 2025. PMID: 40646715
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

