Kinetics of Phase Transitions in Amorphous Carbamazepine: From Sub- Tg Structural Relaxation to High-Temperature Decomposition
- PMID: 40649912
- PMCID: PMC12250088
- DOI: 10.3390/ijms26136136
Kinetics of Phase Transitions in Amorphous Carbamazepine: From Sub- Tg Structural Relaxation to High-Temperature Decomposition
Abstract
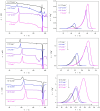
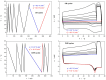
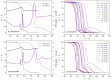
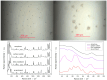
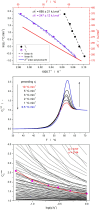
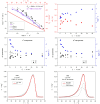
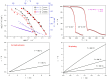
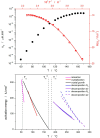
Thermokinetic characterization of amorphous carbamazepine was performed utilizing non-isothermal differential scanning calorimetry (DSC) and thermogravimetry (TGA). Structural relaxation of the amorphous matrix was described in terms of the Tool-Narayanaswamy-Moynihan model with the following parameters: Δh* ≈ 200-300 kJ·mol-1, β = 0.57, x = 0.44. The crystallization of the amorphous phase was modeled using complex Šesták-Berggren kinetics, which incorporates temperature-dependent activation energy and degree of autocatalysis. The activation energy of the crystal growth was determined to be >320 kJ·mol-1 at the glass transition temperature (Tg). Owing to such a high value, the amorphous carbamazepine is stable at Tg, allowing for extensive processing of the amorphous phase (e.g., self-healing of the quench-induced mechanical defects or internal stress). A discussion was conducted regarding the converse relation between the activation energies of relaxation and crystal growth, which is possibly responsible for the absence of sub-Tg crystal growth modes. The high-temperature thermal decomposition of carbamazepine proceeds via multistep kinetics, identically in both an inert and an oxidizing atmosphere. A complex reaction mechanism, consisting of a series of consecutive and competing reactions, was proposed to explain the second decomposition step, which exhibited a temporary mass increase. Whereas a negligible degree of carbamazepine degradation was predicted for the temperature characteristic of the pharmaceutical hot-melt extrusion (~150 °C), the degradation risk during the pharmaceutical 3D printing was calculated to be considerably higher (1-2% mass loss at temperatures 190-200 °C).
Keywords: carbamazepine; crystal growth; structural relaxation; thermal decomposition.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
-
- Cornett E.M., Amarasinghe S.N., Angelette A., Abubakar T., Kaye A.M., Kaye A.D., Neuchat E.E., Urits I., Viswanath O. Valtoco® (Diazepam nasal spray) for the acute treatment of intermittent stereotypic episodes of frequent seizure activity. Neurol. Int. 2021;13:64–78. doi: 10.3390/neurolint13010007. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

