This is a preprint.
Presynaptic vesicles supply membrane for axonal bouton enlargement during LTP
- PMID: 40654927
- PMCID: PMC12247634
- DOI: 10.1101/2025.04.29.651313
Presynaptic vesicles supply membrane for axonal bouton enlargement during LTP
Abstract
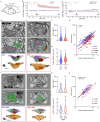
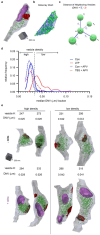
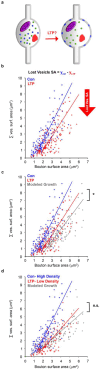
Long-term potentiation (LTP) induces presynaptic bouton enlargement and a reduction in the number of synaptic vesicles. To understand the relationship between these events, we performed 3D analysis of serial section electron micrographs in rat hippocampal area CA1, 2 hours after LTP induction. We observed a high vesicle packing density in control boutons, contrasting with a lower density in most LTP boutons. Notably, the summed membrane area of the vesicles lost in low-density LTP boutons is comparable to the surface membrane required for the observed bouton enlargement when compared to high-density control boutons. These novel findings suggest that presynaptic vesicle density provides a new structural indicator of LTP that supports a local mechanism of bouton enlargement.
Figures



References
-
- Bliss T. & Gardner-Medwin A. Long-lasting potentiation of synaptic transmission in the dentate area of the unanaesthetized rabbit following stimulation of the perforant path. https://physoc.onlinelibrary.wiley.com/doi/epdf/10.1113/jphysiol.1973.sp... (1973) doi: 10.1113/jphysiol.1973.sp010274. - DOI - DOI - PMC - PubMed
-
- Neher E. & Brose N. Dynamically Primed Synaptic Vesicle States: Key to Understand Synaptic Short-Term Plasticity. Neuron 100, 1283–1291 (2018). - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
