This is a preprint.
Use of preclinical Alzheimer's disease trajectories for clinical trial design
- PMID: 40657310
- PMCID: PMC12248167
- DOI: 10.1101/2025.05.01.25326668
Use of preclinical Alzheimer's disease trajectories for clinical trial design
Abstract
Introduction: This study uses longitudinal amyloid biomarker and cognitive data to generate sample size estimates for two-armed, pre-clinical amyloid clearance clinical trials.
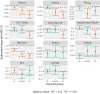
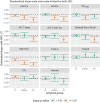
Methods: PET PiB DVR ranges defined three amyloid groups (positive, "A+"; sub threshold/low positive, "subA+"; and negative, "A-") in cognitively unimpaired Wisconsin Registry for Alzheimer's Prevention participants. Amyloid group trajectories estimated from mixed effects models informed per-treatment-arm sample size estimates to detect plausible treatment effects over 3-year (biomarker) or 6-year (cognition) study windows (80% power).
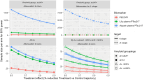
Results: To detect ≥60% slowing in PiB accumulation, ≤40 may be needed per arm for both SubA+ and A+; to detect the same effect sizes in plasma p-tau217 trajectories, ~50-1700 are needed, depending on assay and amyloid subgroup. Among cognitive outcomes, Digit Symbol Substitution and a 5-test Preclinical Alzheimer's Cognitive Composite consistently required fewest (<2000) per arm.
Discussion: Early intervention study planning will benefit from selection of outcomes that are most sensitive to AD biomarker-related preclinical change.
Keywords: Alzheimer’s disease; biomarkers; clinical trial design; preclinical; preclinical intervention.
Conflict of interest statement
Conflict of Interest Statement/Disclosures Authors REL, DLN, KAC, LD, EMJ, RW, RERR, BH have nothing to disclose. HZ has served at scientific advisory boards and/or as a consultant for Abbvie, Acumen, Alector, Alzinova, ALZpath, Amylyx, Annexon, Apellis, Artery Therapeutics, AZTherapies, Cognito Therapeutics, CogRx, Denali, Eisai, Enigma, LabCorp, Merry Life, Nervgen, Novo Nordisk, Optoceutics, Passage Bio, Pinteon Therapeutics, Prothena, Quanterix, Red Abbey Labs, reMYND, Roche, Samumed, Siemens Healthineers, Triplet Therapeutics, and Wave, has given lectures sponsored by Alzecure, BioArctic, Biogen, Cellectricon, Fujirebio, Lilly, Novo Nordisk, Roche, and WebMD, and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program (outside submitted work). SCJ serves as an advisor to Enigma Biomedical and ALZpath.
Figures



References
-
- Payton NM, Marseglia A, Grande G, Fratiglioni L, Kivipelto M, Bäckman L, et al. Trajectories of cognitive decline and dementia development: A 12‐year longitudinal study. Alzheimer’s & Dementia. 2023;19(3):857–67 1552–5260. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
