S100a4+ alveolar macrophages accelerate the progression of precancerous atypical adenomatous hyperplasia by promoting the angiogenic function regulated by fatty acid metabolism
- PMID: 40658605
- PMCID: PMC12259021
- DOI: 10.7554/eLife.101731
S100a4+ alveolar macrophages accelerate the progression of precancerous atypical adenomatous hyperplasia by promoting the angiogenic function regulated by fatty acid metabolism
Abstract
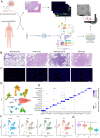
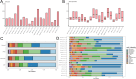
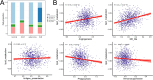
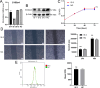
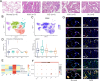
Lung cancer is preceded by premalignant lesions, and what factors drive this transformation and the potential regulatory mode in the context of tumor initiation remain to be elucidated. In the course of precancerous lesions in mice, we found a phasic shift in metabolic patterns. Macrophages are a heterogeneous cell population with high plasticity in the tumor microenvironment. Single-cell interaction and metabolic analyses highlighted a cellular state, S100a4+ alveolar macrophages, which exhibited distinct fatty acid metabolic activity, such as palmitic acid metabolism, at the atypical adenomatous hyperplasia stage, accompanied by an angiogenic-promoting function in a pre-neoplastic setting of mice. These findings were reproducible in human single-cell transcriptomes and had been confirmed by histopathological staining and in vitro cell coculture assays. Taken together, the results from this study demonstrated that the S100a4+ alveolar macrophage subset contributes to tumorigenesis by altering its metabolic state, suggesting that metabolic interventions targeting this cell state in the early stage of disease may delay neoplastic transformation of the lung epithelium.
Keywords: alveolar macrophage; angiogenesis; atypical adenomatous hyperplasia; cancer biology; fatty acid metabolism; human; lung cancer; mouse; precancerous lesion.
© 2024, Huang, Yang et al.
Conflict of interest statement
HH, YY, QZ, YY, ZX, SM, TS, YW, ZL, HB, LZ, LZ No competing interests declared
Figures








Update of
- doi: 10.1101/2024.10.17.618873
- doi: 10.7554/eLife.101731.1
- doi: 10.7554/eLife.101731.2
Similar articles
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of paclitaxel, docetaxel, gemcitabine and vinorelbine in non-small-cell lung cancer.Health Technol Assess. 2001;5(32):1-195. doi: 10.3310/hta5320. Health Technol Assess. 2001. PMID: 12065068
-
[Volume and health outcomes: evidence from systematic reviews and from evaluation of Italian hospital data].Epidemiol Prev. 2013 Mar-Jun;37(2-3 Suppl 2):1-100. Epidemiol Prev. 2013. PMID: 23851286 Italian.
-
M1 Macrophage-Derived TNF-α Promotes Pancreatic Cancer Ferroptosis Via p38 MAPK-ACSL4 Pathway.Curr Mol Med. 2025 Jul 10. doi: 10.2174/0115665240374551250630075409. Online ahead of print. Curr Mol Med. 2025. PMID: 40653839
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
References
-
- Anderson AN, Conley P, Klocke CD, Sengupta SK, Robinson TL, Fan Y, Jones JA, Gibbs SL, Skalet AH, Wu G, Wong MH. Analysis of Uveal Melanoma scRNA Sequencing Data Identifies Neoplastic-Immune Hybrid Cells That Exhibit Metastatic Potential. bioRxiv. 2023 doi: 10.1101/2023.10.24.563815. - DOI
-
- Barton NW, Brady RO, Dambrosia JM, Di Bisceglie AM, Doppelt SH, Hill SC, Mankin HJ, Murray GJ, Parker RI, Argoff CE. Replacement therapy for inherited enzyme deficiency--macrophage-targeted glucocerebrosidase for Gaucher’s disease. The New England Journal of Medicine. 1991;324:1464–1470. doi: 10.1056/NEJM199105233242104. - DOI - PubMed
-
- Bian Y, Li W, Kremer DM, Sajjakulnukit P, Li S, Crespo J, Nwosu ZC, Zhang L, Czerwonka A, Pawłowska A, Xia H, Li J, Liao P, Yu J, Vatan L, Szeliga W, Wei S, Grove S, Liu JR, McLean K, Cieslik M, Chinnaiyan AM, Zgodziński W, Wallner G, Wertel I, Okła K, Kryczek I, Lyssiotis CA, Zou W. Cancer SLC43A2 alters T cell methionine metabolism and histone methylation. Nature. 2020;585:277–282. doi: 10.1038/s41586-020-2682-1. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

