Mitochondrial accumulation of GRK2 as a protective mechanism against hypoxia-induced endothelial dysfunction
- PMID: 40659632
- PMCID: PMC12259972
- DOI: 10.1038/s41420-025-02628-0
Mitochondrial accumulation of GRK2 as a protective mechanism against hypoxia-induced endothelial dysfunction
Abstract
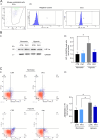
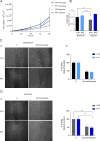
Hypoxia, a condition characterized by a temporary lack of oxygen, causes mitochondrial damage, which in turn leads to endothelial dysfunction. G-protein-coupled receptor kinase 2 (GRK2) plays a key role in vascular homeostasis and remodeling, influencing endothelial function through various pathways. GRK2 moves within the cellular compartments and is linked to mitochondrial function and biogenesis, promoting ATP production and protecting against oxidative stress and cell death. The present study examined how mitochondrial GRK2 accumulation affects vascular reactivity and endothelial function in transient hypoxic conditions. Using a cloning strategy, we employed a small peptide (10aa) TAT-conjugated based on the pleckstrin homology domain of GRK2 to redirect GRK2 from the plasma membrane to the mitochondria. Mitochondrial accumulation of GRK2 increases vasodilatory responses in isolated swine artery segments, indicating potential therapeutic applications for cardiovascular disorders. Furthermore, in endothelial cells, GRK2 accumulation within mitochondria protects membrane potential, mitochondrial mass and prevents oxidative damage and cell death caused by transient hypoxia. Our findings show that GRK2 accumulation in mitochondria represents a potential therapeutic target to prevent transient hypoxia-induced damage.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors have no conflicts of interest to declare. Ethics approval and consent to participate: All animal care and experimental procedures were conducted according to the principles of the “Guide for the Care and Use of Laboratory Animals” [48] in accordance with the Italian national law (Legislative Decree 26/2014) and the recommendations of the European Community (63/2010/CEE), and have been approved by the Italian Ministry of Health for the animal facility authorization protocol number AUT.N. 211/2019-PR (for studies in mice) and protocol number AUT.N. 142/2022-PR (for studies in pigs). No human subjects were included in this study.
Figures




References
LinkOut - more resources
Full Text Sources

