Mitochondrial proteomic adaptations to daily torpor in the Djungarian hamster (Phodopus sungorus)
- PMID: 40663131
- PMCID: PMC12367877
- DOI: 10.1007/s00360-025-01625-0
Mitochondrial proteomic adaptations to daily torpor in the Djungarian hamster (Phodopus sungorus)
Abstract
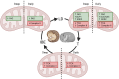
Hibernation is an adaptive strategy that conserves energy in response to environmental challenges. While mitochondrial proteomic adaptations are well-documented in deep hibernators, the proteomic changes underlying daily torpor remain less clear. We investigated mitochondrial proteomic adaptations in the liver of a daily hibernator, the Djungarian hamster (Phodopus sungorus), across different hibernation phases. Hamsters were maintained under long-day (summer) or short-day photoperiods (winter), to induce torpor. Livers from summer, torpor, and interbout euthermia phases were analyzed by liquid chromatography-mass spectrometry with labelled standards of mitochondrial energy metabolism proteins, resulting in accurate quantitative proteomics. Differential protein regulation was assessed using empirical Bayes models with false discovery rate correction. Increased abundance of fatty acid oxidation enzymes during hibernation indicates a seasonal metabolic shift toward lipid utilization, similar to deep hibernators. Additionally, torpor featured elevated complex II subunits and tricarboxylic acid cycle enzymes representing evolutionary adaptations specific to daily torpor, likely to cater higher energy demands necessary to maintain torpid body temperature above 15 °C in near-freezing ambient temperatures. This represents evolutionary adaptations specific to daily torpor. Increased levels of the mitochondrial uncoupling-related solute carrier family 25 member 5 (SLC25A5) may be responsible for both thermogenesis and limiting production of reactive oxygen species. Furthermore, the selective upregulation of SOD2 during torpor underscores its critical role in mitigating reactive oxygen species accumulation during metabolic transitions. In summary, daily torpor exhibits unique mitochondrial proteomic adaptations that distinguish it from deep torpor, which may be necessary to enable torpor at body temperatures well above the ambient temperature.
Keywords: Daily torpor; Djungarian hamster; Energy metabolism; Hibernation; Mitochondrial proteomics.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethical approval: All animal work was licensed under the Animals (Scientific Procedures) Act of 1986 and approved by the University of Aberdeen, Rowett Institute for Nutrition and Health ethics committee. Competing interests: The authors have no relevant financial or non-financial interests to disclose.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

