This is a preprint.
Pharmacological inhibition of G protein-coupled receptor kinase 5 decreases high-fat diet-induced hepatic steatosis in mice
- PMID: 40666930
- PMCID: PMC12262720
- DOI: 10.1101/2025.06.25.661655
Pharmacological inhibition of G protein-coupled receptor kinase 5 decreases high-fat diet-induced hepatic steatosis in mice
Update in
-
Pharmacological inhibition of G protein-coupled receptor kinase 5 decreases high-fat diet-induced hepatic steatosis in mice.Biochem Biophys Res Commun. 2026 Jan 8;795:153078. doi: 10.1016/j.bbrc.2025.153078. Epub 2025 Dec 3. Biochem Biophys Res Commun. 2026. PMID: 41353922
Abstract
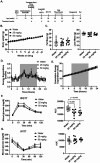
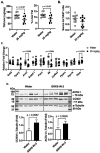
G protein-coupled receptor kinase 5 (GRK5) is implicated in the pathogenesis of obesity in both humans and rodent models. Our previous work demonstrated that genetic deletion or pharmacological inhibition of GRK5 suppresses 3T3-L1 adipocyte differentiation. Here, we assessed the small-molecule GRK5 inhibitor, GRK5-IN-2, for its effects on metabolic tissues and therapeutic potential in a diet-induced obesity mouse model. Mice were fed a high-fat diet for 8 weeks to induce obesity, followed by continued a high-fat diet with oral administration of GRK5-IN-2 (25 or 50 mg/kg) or water vehicle, five days per week for an additional 16 weeks. GRK5-IN-2 treatment had no effect on body weight, fat/lean mass, insulin tolerance, food intake, or energy expenditure but significantly reduced hepatic triglyceride accumulation and de novo lipogenesis. A follow-up study using 25 mg/kg of GRK5-IN-2 confirmed no effect on adiposity but reduced hepatic triglycerides. GRK5-IN-2 treatment decreased expression of the lipogenic gene Acc2 while upregulating lipid utilization proteins COXIV and ACSL1 in the liver, likely contributing to lower triglyceride levels. Together, these findings suggest that GRK5 inhibition selectively modulates hepatic lipid metabolism without altering systemic metabolic parameters, highlighting GRK5 as a potential therapeutic target for fatty liver disease.
Keywords: Adiposity; G protein-coupled receptor kinase 5; Hepatic steatosis; High fat diet; Obesity.
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures



References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
