This is a preprint.
Synaptotagmin 1 and Synaptotagmin 7 promote MR1-mediated presentation of Mycobacterium tuberculosis antigens
- PMID: 40667066
- PMCID: PMC12262809
- DOI: 10.1101/2025.06.23.660389
Synaptotagmin 1 and Synaptotagmin 7 promote MR1-mediated presentation of Mycobacterium tuberculosis antigens
Abstract
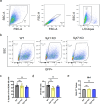
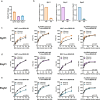
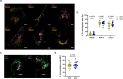
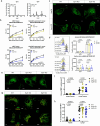
Mycobacterium tuberculosis (Mtb) is an intracellular pathogen that can be sensed by T cells, which are essential for the control of infection. In comparison to viral infections, Mtb antigens are relatively rare and hence, challenging to sample. Specialized antigen presentation pathways enable the presentation of such scarce antigens to CD8+ T cells, which are, thus, uniquely poised to survey intracellular environments. A subset of CD8+ T cells prevalent in the airways, known as mucosal associated invariant T (MAIT) cells, can be activated through the presentation of Mtb antigens via the MHC class I-related protein 1 (MR1) molecule. Prior work demonstrates that endosomal calcium signaling is critical for MR1-mediated presentation of Mtb-derived antigens. Here, we show that the calcium-sensing trafficking proteins Synaptotagmin (Syt) 1 and Syt7 specifically promote MAIT cell activation in response to Mtb-infected cells. In bronchial epithelial cells, Syt1 and Syt7 localize to late endo-lysosomes and MR1 vesicles. Loss of Syt1 and Syt7 results in enlarged MR1 vesicles and an increased number of MR1 vesicles in close proximity to Mtb-containing vacuoles during infection. This study identifies a novel pathway in which Syt1 and Syt7 facilitate the translocation of MR1 from Mtb-containing vacuoles, potentially to the cell surface for antigen presentation.
Conflict of interest statement
Competing interests The authors declare no competing interests.
Figures


Similar articles
-
Two-pore channels in MR1-dependent presentation of Mycobacterium tuberculosis infection.PLoS Pathog. 2025 Aug 4;21(8):e1013342. doi: 10.1371/journal.ppat.1013342. eCollection 2025 Aug. PLoS Pathog. 2025. PMID: 40758737 Free PMC article.
-
Calcium Signaling in MR1-Dependent Antigen Presentation of Mycobacterium tuberculosis.Res Sq [Preprint]. 2023 Aug 30:rs.3.rs-3154465. doi: 10.21203/rs.3.rs-3154465/v1. Res Sq. 2023. Update in: PLoS Pathog. 2025 Aug 4;21(8):e1013342. doi: 10.1371/journal.ppat.1013342. PMID: 37693580 Free PMC article. Updated. Preprint.
-
Modulation of riboflavin biosynthesis and utilization in mycobacteria.Microbiol Spectr. 2024 Aug 6;12(8):e0320723. doi: 10.1128/spectrum.03207-23. Epub 2024 Jun 25. Microbiol Spectr. 2024. PMID: 38916330 Free PMC article.
-
Unconventional T cells in anti-cancer immunity.Front Immunol. 2025 Jul 17;16:1618393. doi: 10.3389/fimmu.2025.1618393. eCollection 2025. Front Immunol. 2025. PMID: 40746558 Free PMC article. Review.
-
Signals that control MAIT cell function in healthy and inflamed human tissues.Immunol Rev. 2024 May;323(1):138-149. doi: 10.1111/imr.13325. Epub 2024 Mar 22. Immunol Rev. 2024. PMID: 38520075 Free PMC article. Review.
References
-
- Global Tuberculosis Report 2024. (World Health Organization, Geneva, 2024).
-
- Pai M. et al. Tuberculosis. Nat. Rev. Dis. Primer 2, 16076 (2016). - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
