This is a preprint.
Hydraulic Activation of the AsLOV2 photoreceptor
- PMID: 40667302
- PMCID: PMC12262554
- DOI: 10.1101/2025.06.19.660617
Hydraulic Activation of the AsLOV2 photoreceptor
Abstract
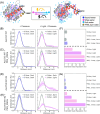
How proteins transduce environmental signals into mechanical motion remains a central question in biology. This study tests the hypothesis that blue light activation of AsLOV2 gives rise to concerted water movement that induce protein conformational extensions. Using electron and nuclear magnetic resonance spectroscopy, along with atomistic molecular dynamics simulations at high pressure, we find that activation, whether initiated by blue light or high pressure, is accompanied by selective expulsion of low-entropy, tetrahedrally coordinated "wrap" water from hydrophobic regions of the protein. These findings suggest that interfacial water serves as functional constituents to help reshape the protein's free energy landscape during activation. Our study highlights hydration water as an active medium with the capacity to drive long-range conformational changes underlying protein mechanics and offers a new conceptual understanding for engineering externally controllable protein actuators for biomedical studies to smart materials.
Figures




References
-
- Jentzsch K., Wirtz A., Circolone F., Drepper T., Losi A., Gärtner W., Jaeger K.-E., Krauss U., Mutual Exchange of Kinetic Properties by Extended Mutagenesis in Two Short LOV Domain Proteins from Pseudomonas putida. Biochemistry 48, 10321–10333 (2009). - PubMed
-
- Kasahara M., Swartz T. E., Olney M. A., Onodera A., Mochizuki N., Fukuzawa H., Asamizu E., Tabata S., Kanegae H., Takano M., Christie J. M., Nagatani A., Briggs W. R., Photochemical Properties of the Flavin Mononucleotide-Binding Domains of the Phototropins from Arabidopsis, Rice, andChlamydomonas reinhardtii. Plant Physiology 129, 762–773 (2002). - PMC - PubMed
-
- Chaves I., Pokorny R., Byrdin M., Hoang N., Ritz T., Brettel K., Essen L.-O., van der Horst G. T. J., Batschauer A., Ahmad M., The Cryptochromes: Blue Light Photoreceptors in Plants and Animals. Annual Review of Plant Biology 62, 335–364 (2011). - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
