Metabolic Dysfunction-Associated Steatohepatitis Diagnosis and Management in Germany: Insights From an Expert Consensus Panel
- PMID: 40668568
- PMCID: PMC12266315
- DOI: 10.1111/liv.70225
Metabolic Dysfunction-Associated Steatohepatitis Diagnosis and Management in Germany: Insights From an Expert Consensus Panel
Abstract
Background: Metabolic dysfunction-associated steatotic liver disease (MASLD) is a major cause of chronic liver disease. Metabolic dysfunction-associated steatohepatitis (MASH), a progressive form of MASLD, can lead to fibrosis and cirrhosis. The incidence and burden of MASH in Germany are expected to double by 2030, while diagnostic and management challenges persist. Expert consensus on diagnostic strategies and treatment modalities in MASLD and MASH is required.
Objectives: The panel aimed to gather insights and consensus on the diagnostic pathway and current treatment modalities for MASH in Germany.
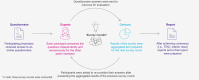
Methods: A three-round web-based survey, integrating Delphi panel methodology with a standard survey, was conducted from February to May 2024 to reach consensus on predefined questions. The survey involved 12 gastroenterology, diabetology and/or hepatology specialists in Germany.
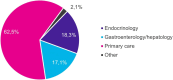
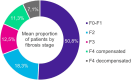
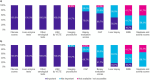
Results: The Delphi panel revealed that ~75% of MASH patients in Germany remain undiagnosed. Non-invasive measures, such as fibrosis scores (values from clinical and imaging tests to assess fibrosis), liver enzyme tests and liver stiffness measurement, were primary methods for diagnosing and monitoring MASH patients. Lifestyle modifications were the primary management strategy, given the absence of approved pharmacological treatments for MASH. The panel also highlighted significant challenges in managing MASH, including the lack of approved medications and the difficulty in sustaining lifestyle changes.
Conclusions: The survey underscores the substantial underdiagnosis of MASH and the reliance on non-invasive diagnostic methods in Germany. The lack of approved treatments necessitates a focus on lifestyle modifications and comorbidity management. The Delphi panel's insights call for enhanced screening, early detection and standardised algorithms to improve patient outcomes.
Keywords: MASH; MASLD; NAFLD; NASH; non‐invasive tests.
© 2025 The Author(s). Liver International published by John Wiley & Sons Ltd.
Conflict of interest statement
Y.K., P.R. and T.R. are employees of Madrigal Pharmaceuticals Inc. and own stock. J.S.H. is an employee of PharmaLex GmbH (former Xcenda GmbH). PharmaLex GmbH (former Xcenda GmbH) received consulting fees for the execution of the study and for the manuscript preparation from Madrigal Pharmaceuticals Inc. H.B. received consultancy fees from Advanz Pharma, Echosens, Gilead Sciences, GSK, Intercept, Ipsen, Roche and received speaker honoraria from Falk Foundation, Gilead, Advanz and GSK. W.‐P.H. receives consultancy fees from AbbVie, Gilead Sciences, Ipsen Pharma, Falk Pharma and Madrigal Pharmaceuticals Inc. E.R. has received honoraria for consulting or lectures from AbbVie, Amgen, Intercept, Madrigal, Medac, Merz, Norgine, Falk Foundation, Gilead, Pfizer, Repha and Takeda. J.M.S. receives consultancy fees from Akero, Alentis, Alexion, Altimmune, AstraZeneca, 89Bio, Bionorica, Boehringer Ingelheim, Gilead Sciences, GSK, HistoIndex, Ipsen, Inventiva Pharma, Madrigal Pharmaceuticals, Kríya Therapeutics, Lilly, MSD Sharp & Dohme GmbH, Novartis, Novo Nordisk, Pfizer, Roche, Sanofi and Siemens Healthineers; speaker honorarium from AbbVie, Boehringer Ingelheim, Gilead Sciences, Ipsen, Novo Nordisk and Madrigal Pharmaceuticals. Stockholder options: Hepta Bio. N.S. receives consultancy fees from Boehringer Ingelheim, Madrigal Pharmaceuticals Inc., Pfizer and Lilly. K.D. receives a consultancy fee, travel support and a research grant from Gilead and lecture fees from Alnylam. F.T.'s lab has received research grants (funding to the institution) from AstraZeneca, MSD and Gilead. F.T. has received honoraria for consulting or lectures from Madrigal, Gilead, AbbVie, Falk, AstraZeneca, Boehringer, MSD, GSK, Ipsen, Pfizer, Novartis, Novo Nordisk and Sanofi. J.W. is a lecturer and advisory board member for AbbVie, Gilead, Intercept/Advanz Pharma, GSK, Ipsen and Novo Nordisk; research grants from AbbVie and Gilead. A.G. reports support from the IMI2 LITMUS project; research grants from Novartis, Falk and Intercept; consulting or speaker's fees from AbbVie, Advanz, Albireo, Alexion, AstraZeneca, Bayer, BMS, Boehringer, Burgerstein, CSL Behring, Eisai, Falk, Gilead, Heel, Intercept, Ipsen, Merz, MSD, Novartis, Novo Nordisk, Orphalan, Pfizer, Roche and Sanofi‐Aventis; and travel/meeting support from Intercept, Gilead, AbbVie and Falk. A.P. participates in advisory boards for AbbVie, Gilead, Xcenda and Madrigal, as well as speaker fees from AbbVie, BMS, Gilead, MSD and Novo Nordisk. A.G. is an advisory board or steering committee member for AbbVie, Advanz, Albireo, Alexion, AstraZeneca, Bayer, Bristol Myers Squibb, CSL Behring, Eisai, Falk, Gilead, Heel, Intercept, Ipsen, Merz, MSD, Novartis, Novo Nordisk, Pfizer, Roche, Sanofi‐Aventis and Sequana, and a speaker for Orphalan. K.‐G.S. received consultancy fees from AbbVie, Gilead and Falk.
Figures
References
-
- Tacke F., Horn P., Wai‐Sun Wong V., et al., “EASL–EASD–EASO Clinical Practice Guidelines on the Management of Metabolic Dysfunction‐Associated Steatotic Liver Disease (MASLD),” Journal of Hepatology 81 (2024): 492–542. - PubMed
-
- Roeb E., Canbay A., Bantel H., et al., “Amendment ‘Neue Nomenklatur zur MASLD (Metabolic Dysfunction Associated Steatotic Liver Disease; Metabolische Dysfunktion Assoziierte Steatotische LEBERERKRANKUNG)’ Zur S2k‐Leitlinie ‘Nicht‐Alkoholische Fettlebererkrankung’ (v.2.0/April 2022) der Deutschen Gesellschaft für Gastroenterologie Verdauungs‐ und Stoffwechselkrankheiten (DGVS),” Zeitschrift Für Gastroenterologie 62, no. 7 (2024): 1077–1087. - PubMed
-
- European Association for the Study of the Liver , European Association for the Study of Diabetes , and European Association for the Study of Obesity , “EASL‐EASD‐EASO Clinical Practice Guidelines for the Management of Non‐Alcoholic Fatty Liver Disease,” Journal of Hepatology 64, no. 6 (2016): 1388–1402. - PubMed
-
- Roeb E., Canbay A., Bantel H., et al., “Aktualisierte S2k‐Leitlinie Nicht‐Alkoholische Fettlebererkrankung der Deutschen Gesellschaft für Gastroenterologie, Verdauungs‐ und Stoffwechselkrankheiten (DGVS) – April 2022 – AWMF‐Registernummer: 021–025,” Zeitschrift für Gastroenterologie 60, no. 9 (2022): 1346–1421. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

