Interleukin-4 prevents increased endothelial permeability by inducing pericyte survival and modulating microglial responses in diabetic retinopathy
- PMID: 40671907
- PMCID: PMC12263392
- DOI: 10.3389/fendo.2025.1609796
Interleukin-4 prevents increased endothelial permeability by inducing pericyte survival and modulating microglial responses in diabetic retinopathy
Abstract
Introduction: Retinal vascular leakage due to increased endothelial permeability is a major contributor to the pathogenesis of diabetic retinopathy (DR) and visual impairment. Pericyte loss and microglia-mediated inflammation exacerbate this vascular dysfunction. Interleukin-4 (IL-4) is known for its anti-inflammatory and tissue-protective properties, but its role in DR remains unclear.
Methods: We evaluated IL-4 expression and signaling in the retinas of streptozotocin-induced diabetic mice. In vitro assays were conducted under high-glucose and TNF-α conditions using retinal endothelial cells, pericytes, and microglia to assess IL-4's effects on barrier function, cell viability, and inflammatory state. Pathway-specific analyses were performed to investigate PI3K/AKT and STAT6 signaling.
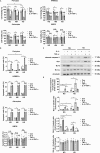
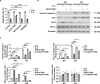
Results: IL-4 expression and downstream signaling were significantly reduced in diabetic retinas. IL-4 promoted pericyte survival via PI3K/AKT activation and modulated microglial functional profiles through STAT6 signaling, favoring an anti-inflammatory phenotype. These effects contributed to restored endothelial barrier integrity and tight junction protein expression under diabetic stress conditions in vitro.
Conclusion: IL-4 supports retinal vascular stabilization in DR by preserving pericyte viability and modulating microglial activity. These findings highlight IL-4 as a potential therapeutic target for preventing or slowing DR progression and warrant further preclinical investigation.
Keywords: diabetic retinopathy; endothelial permeability; interleukin-4; microglia functional states; pericytes; signal transducer and activator of transcription 6.
Copyright © 2025 Yun.
Conflict of interest statement
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





Similar articles
-
Inhibition of Dectin-1 alleviates inflammation in early diabetic retinopathy by regulating microglia phenotype.Gene. 2025 Sep 5;963:149572. doi: 10.1016/j.gene.2025.149572. Epub 2025 May 15. Gene. 2025. PMID: 40381970
-
Interleukin-1β induces pericyte apoptosis via the NF-κB pathway in diabetic retinopathy.Biochem Biophys Res Commun. 2021 Mar 26;546:46-53. doi: 10.1016/j.bbrc.2021.01.108. Epub 2021 Feb 8. Biochem Biophys Res Commun. 2021. PMID: 33571904
-
Microglia Promote Endothelial Cell Activation Through NSUN2-Mediated SQLE m5C Modification in Diabetic Retinopathy.FASEB J. 2025 Jun 30;39(12):e70742. doi: 10.1096/fj.202500302RR. FASEB J. 2025. PMID: 40536094
-
[Mouse models of diabetic retinopathy: systematic review of the literature].J Fr Ophtalmol. 2013 Mar;36(3):268-76. doi: 10.1016/j.jfo.2012.08.001. Epub 2013 Feb 22. J Fr Ophtalmol. 2013. PMID: 23434332 French.
-
Fenofibrate for diabetic retinopathy.Cochrane Database Syst Rev. 2023 Jun 13;6(6):CD013318. doi: 10.1002/14651858.CD013318.pub2. Cochrane Database Syst Rev. 2023. PMID: 37310870 Free PMC article.
References
-
- Lam NV. Adult eye conditions: diabetic retinopathy and age-related macular degeneration. FP Essent. (2022) 519:24–8. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

