PIONOCONUS: A PISCIVOROUS SUBGENUS OF CONUS GASTROPODS
- PMID: 40672043
- PMCID: PMC12266717
- DOI: 10.4002/040.067.0104
PIONOCONUS: A PISCIVOROUS SUBGENUS OF CONUS GASTROPODS
Abstract
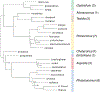
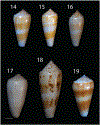
Alan Kohn showed that two cone-snail species-Conus striatus Linnaeus, 1758, and Conus catus Hwass, 1792-hunt fish as their primary prey. In the seven decades since then, it has been established that these two species belong to the subgenus Pionoconus Mörch, 1852, a well-defined lineage of Conus Linnaeus, 1758, and one of eight piscivorous cone-snail clades. In this review, an integrated multidisciplinary framework for the biology of Pionoconus is presented, based on the numerous research papers published since Kohn's seminal discovery. The molecular biology, phylogeny, biogeography, life history, and prey-capture strategy of Pionoconus are among the topics reviewed, along with more specialized subjects, e.g., human envenomation, culinary recipes, shell-collecting history, and biomedical applications. These illustrate the continuing impact of Kohn's scientific contributions.
Keywords: Pionoconus; clades; conotoxins; envenomation; taxonomy.
Figures





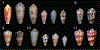


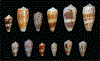
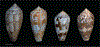

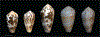
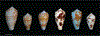
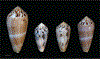


References
-
- Akondi KB, Muttenthaler M, Dutertre S, Kaas Q, Craik DJ, Lewis RJ, & Alewood PF, 2014, Discovery, synthesis, and structure-activity relationships of conotoxins. Chemical Reviews, 114(11): 5815–5847, https://pubs.acs.org/doi/10.1021/cr400401e. - DOI - PMC - PubMed
-
- Ali JR, & Aitchison JC, 2008, Gondwana to Asia: plate tectonics, paleogeography and the biological connectivity of the Indian sub-continent from the Middle Jurassic through latest Eocene (166–35 Ma). Earth-Science Reviews, 88(3): 145–166, 10.1016/j.ear-scirev.2008.01.007. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
